Combining ‘assembly line’ synthesis, spectroscopy and computational predictions solves stereochemistry riddle
After failing to make the superbug-fighting molecule baulamycin, UK researchers have overturned the chemical structure it was originally assigned.1 Varinder Aggarwal and colleagues at the University of Bristol had been trying to make both baulamycin A and B using the ‘assembly line synthesis’ approach they developed. But the method would also provide answers to a challenge arising from the baulamycins’ exotic origins.
When researchers including David Sherman from the University of Michigan, US, discovered the baulamycins in marine bacteria from off Costa Rica they obtained a few milligrams of each.2 This was enough to show they could halt the growth of methicillin-resistant Staphylococcus aureus (MRSA) and anthrax, but working out the structure of the molecules’ long, floppy, carbon chain was harder. The chains have seven chiral centres, where surrounding atoms can take one of two mirror-image arrangements, giving 128 different possible stereoisomer options for the correct structure. The only method suitable was NMR, and it is ‘very difficult to assign chiral centres using NMR alone in highly complex structures such as the baulamycins,’ Sherman tells Chemistry World.
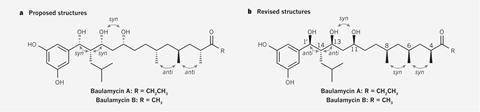
Aggarwal’s assembly line approach can precisely add chiral centres one after another in the correct orientation, making it well-suited to producing baulamycins. But after thinking they’d made baulamycin A, they found their NMR data didn’t match the original samples. Instead, it appeared that the true structure was a different one of the many possible than the team that discovered it had proposed. ‘We were initially surprised, but when you look into the problem more deeply you realise the chances of it being wrong were actually quite high,’ Bristol chemist Eddie Myers tells Chemistry World.
A molecule of two halves
Similar problems have arisen before, Myers adds, but often natural products link around on themselves in relatively rigid rings. Their structures can therefore be solved by predicting NMR parameters for the limited number of conformations ring structures can adopt using computational chemistry. But because baulamycin is floppy, many more conformations are possible, complicating the challenge so severely that the computational approach alone wasn’t sufficient.
Because the Bristol team had split baulamycin A into two halves to make it, rather than synthesising and analysing all 128 structural options, they investigated each half in turn. For one half, they compared the NMR data they’d computed for all possible arrangements with the original molecule. That narrowed the options for the correct relative arrangement for this half of the molecule down to just one.
Yet this method couldn’t distinguish between four arrangements for the other half of the molecule. The researchers therefore turned to synthesis, with the assembly line approach able to precisely produce all four arrangements at once. By introducing mixtures of the two different possible chiral fragments at each of two steps, the team made a four-component mixture. Ensuring that the arrangements were generated in different amounts so they could be easily differentiated by NMR, comparison with the original data identified the correct one.
Having determined the arrangement in each half of the molecule, the team then linked them together in the four remaining ways possible. Again, comparing the different products’ NMR data to the original sample’s indicated which was ultimately correct.
Sherman is ‘very pleased’ that the Bristol chemists have revised the two chiral centres that his team had mis-assigned, calling the work ‘outstanding’. The groups are now working together, with the Michigan team due to evaluate the different baulamycin isomers’ antibiotic power. ‘We want to see how the stereochemistry affects its activity,’ Myers says.
References
- J Wu et al, Nature, 2017, DOI: 10.1038/nature23265
- A Tripathi et al, J. Am. Chem. Soc., 2014, DOI: 10.1021/ja4115924











No comments yet