The chemistry of uranium and the other actinides is reaching beyond nuclear uses, revealing surprising behaviour and capabilities, finds Andy Extance
With their Schlenk lines and gloveboxes, at first glance Steve Liddle’s chemistry labs at the University of Nottingham in the UK are much like any other. But among the familiar features are signs that might raise alarm, even among chemists used to dealing with experimental hazards. Researchers wearing personal dosimeters hint at the labs’ most unusual feature: the locked safe, and the uranium stockpile inside.

Liddle’s team is part of a new generation exploring chemistry on the wild frontier at the bottom of the periodic table, among the radioactive actinide elements. Thanks to the large nuclear mass of uranium, plutonium and their fellows, it’s a place where relativity and quantum mechanics wrestle to direct bonding and reactivity. In many cases, chemists simply do not yet know what effects that conflict has. To help clean up existing nuclear waste and prevent future problems as countries pursue nuclear rather than fossil fuel energy, they have renewed efforts to find out. And as they do so, the basic knowledge they are gaining could benefit synthesis, catalysis, data storage and chemistry as a whole.
Scientists initially grouped elements from thorium onwards directly beneath the d-block transition metals, as they had similar oxidation states. But when actinide pioneer Glenn Seaborg proposed they should sit below the lanthanides, he thought that their 5f orbitals would behave differently to 4f electrons in the elements above. In uranium, mass-driven relativistic effects first influence s- and p-orbitals, stabilising them and drawing them nearer to its nucleus. These stabilised orbitals shield d- and f-shell electrons from the nuclear charge, allowing them to spread out.
‘The consequence is that 5f electrons become more available for redox chemistry and bonding,’ Liddle says. ‘In the absence of relativistic effects, you would expect actinide chemistry to be much more like the lanthanides, dominated by the 3+ oxidation state, with very little covalency.’ The resulting lively debate over the nature of actinide bonding helped draw him in. ‘If there is any covalency, what’s its nature? How much f-orbital character is employed and how much d-orbital? What ligand character do you have blending into this? I thought there were some really interesting opportunities in terms of studying novel bonding linkages in uranium.’
New nuclear generation

Its potential for military use and power generation kept actinide research well-funded through the second world war and cold war, but by the 1990s the field’s scientists were rapidly retiring. This could be a problem. ‘Many countries now have a legacy of the cold war,’ says Dave Clark from Los Alamos National Laboratory (LANL) in New Mexico, US. ‘We now have environmental contamination [see box below]; we have nuclear waste that needs to be dealt with. There’s also recognition globally that if nuclear energy is going to move forward, then the nuclear waste problem has to be solved. As a result, we needed to educate and train a whole new generation of technical people who would have the quality and breadth needed to address these complex problems.’ Clark therefore became founding director of the Seaborg Institute at LANL, which partners with universities to develop the skills needed.
Actinides are still the wild west of the periodic table
Polly Arnold
Only around 3% of spent nuclear fuel’s mass is radioactive waste: zinc and all the heavier elements arising from fission. The remainder is potentially useful plutonium and uranium, providing strong motivation to find chemical recycling methods. ‘When you burn nuclear fuels, you generate the periodic table,’ Clark says. ‘A lot of that’s easy to separate, but when you get to the trivalent actinides – heavy ones like americium and curium – their stable states are trivalent and they have the same charge-to-size radius ratio as the lanthanides, so it’s harder.’ But Clark notes Seaborg suggested a solution: exploit actinides’ likely preference, compared to the lanthanides, for covalent bonding.
That challenge led to the creation of the lab Marinella Mazzanti joined at the CEA, the French Alternative Energies and Atomic Energy Commission, in Grenoble in 1996. Over the following decade, her group and researchers worldwide showed overlap between the actinides’ extended 5f orbitals and the electrons donated by ligands could separate them from lanthanides in spent nuclear fuel. ‘There is stronger interaction between actinides and some softer sulfur-, phosphorus- and nitrogen-containing ligands,’ she explains.
Potential ligands identified for this purpose include bis-triazinyl bipyridines and dithiophosphinic acids. But this success, and the falling popularity of nuclear power since the 2011 crisis at the Fukushima nuclear plant in Japan has changed the CEA team’s direction. Like Liddle and many others, Mazzanti now hopes that the actinides will prove a fertile hunting ground for fundamental chemical discoveries.
Race to the bottom row
Despite the powerful causes motivating actinide chemistry research, much remains to be learned, according to Polly Arnold at the University of Edinburgh, UK. ‘It’s still the wild west part of the periodic table,’ she says. ‘It wasn’t until about 15 years ago that we really had a definitive picture of the exact order of how the orbitals fill up with electrons in the uranyl ion [UO22+], which has been around forever. If we didn’t agree on the ordering for uranyl ions until then, you might think we don’t have a hope with anything else.’
As actinides have many orbitals with similar energy levels, it’s also difficult to characterise their compounds using traditional methods like nuclear magnetic resonance (NMR) spectroscopy. Unpaired electrons can surround their nuclei, resulting in paramagnetic compounds in which individual electrons’ magnetic fields shift and broaden NMR resonances. And because for safety’s sake reactions are commonly done on very small scales, there often isn’t enough product to get structural information through crystallography.
While modern scientists still face these problems, today’s instruments can provide information to cross-reference. ‘Our NMR spectrometers are better, we can look at paramagnets so much more easily,’ Arnold says. ‘And it used to be we couldn’t possibly get a crystal structure from the tiny crystals and powders that we fish out of our tubes these days. When you’re working with these big, difficult paramagnets, crystal structures are incredibly helpful. We can work at a pace now that the pioneers couldn’t have.’
Actinide coordination chemistry lags behind the rest of the elements says Karsten Meyer, from Friedrich Alexander University in Erlangen, Germany. While transition metal coordination chemistry has been studied since the late 19th century, similar chemistry in uranium only started evolving noticeably in 1989. ‘Uranium-based transition metal-like coordination chemistry was only seen once Dave Clark and Al Sattelberger published the syntheses for well-defined soluble forms of tetrakis-THF uranium trisiodide and solvate-free uranium trisamide,’ he says.1
These starting materials enabled Meyer’s research, from 1998 as a postdoc at the Massachusetts Institute of Technology in the US, and then from 2001 in his own University of California, San Diego (UCSD) group. ‘Everything we did was new and exciting,’ he recalls. In 2004, his UCSD team showed that carbon dioxide could coordinate to uranium in a previously unseen binding mode: end-on, through an oxygen atom.2 That was the first molecular uranium complex to appear in Science, Meyer says. ‘Since 2004 there’s been another Science paper on molecular uranium chemistry almost every year – uranium chemistry has taken off.’
Avoiding radiophobia
Seeing such strange molecules triggered Liddle’s interest, and he feels the opportunity was amplified by the limited number of scientists working on actinides. ‘I think people have been put off by their reputation,’ he says. He uses depleted uranium, which has below 0.2% 235 U, and is therefore less radioactive than the natural form. It must be carefully tracked, securely stored, and researchers’ exposure monitored through dosimeters, but otherwise conventional inert chemical conditions provide adequate precautions. ‘I don’t think working with depleted uranium, as long as you don’t go to a ridiculous scale, is any worse than tin or lead. There are many more toxic compounds that chemists work with on a routine basis that aren’t tracked. I don’t think there’s the same level of scrutiny or controversy because they’re not radioactive, but they could be infinitely more toxic.’
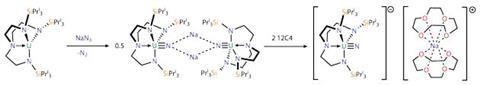
In 2012, Liddle’s team delivered a long-standing target in actinide chemistry: a terminal uranium nitride complex that is stable at room temperature.3 This direct link between uranium and an unsubstituted nitrogen had previously only been seen fleetingly, but was stabilised by a crowded ligand environment. ‘People were beginning to think that this link was inherently unstable. It all came down to a very talented PhD student, exactly the right kind of ligand and the right synthetic approach, and it all just fell into place,’ Liddle says. He points out that many textbook organometallic complexes made with transition metals are yet to be produced with actinides. ‘The d-block organometallics went through a rapid growth phase in the 1960s and 1970s. Uranium and plutonium are going through this phase now, getting what will be future textbook examples made for the first time.’
The main importance of making new actinide molecules is in clarifying their reactivity, according to Arnold. Her team was the first to persuade something to form a covalent bond to an oxygen atom in the uranyl ion, a key intermediate in uranium extraction and processing.4 ‘We block one oxo group, and then found that we were able to reductively silylate the other oxo group,’ she explains. ‘We’ve been able to push on and are now making compounds where the oxo group can be bound to most of the metals in the periodic table.’ These findings seemingly contradict how the uranyl ion has been proposed to react in the environment, a situation Arnold says still confuses and fascinates her. But making these new actinide molecules provides the opportunity to probe them using techniques like x-ray absorption spectroscopy, whose data in turn helps chemists refine their theoretical understanding.
Fighting to clean up a weapons plant

In 1989, the US Federal Bureau of Investigations and Environmental Protection Agency staged an unprecedented dawn raid at the Rocky Flats, Colorado, nuclear weapons production site. Since its opening in 1952, fires, leaks and accidents had released radioactive materials into the surrounding environment. The site was closed down and in 1992 operators Rockwell International pleaded guilty to 10 environmental crimes, agreeing to pay an $18.5 million (£11.3?million) fine. Originally, the US Department of Energy estimated the clean-up would cost over $37 billion and take 70 years.
Without the actinide chemistry knowledge that had been available when Rocky Flats was open, the contractors turned to scientists like LANL’s Dave Clark. The major concern revolved around measurements showing that plutonium levels changed whenever it rained. ‘People thought the plutonium was dissolved and in the groundwater, but plutonium is incredibly insoluble, 10-9 moles per litre, three orders of magnitude below silicon dioxide,’ Clark recalls. ‘Insoluble radionuclides can form colloidal particles, and they can move on the surface by wind dispersion or being moved by the rain. Every time it rained particles would move at the surface. We recognised that the plutonium was only in the upper few centimetres of soil, so it allowed an extensive surface clean-up and a re-vegetation. It was a big success, and saved a lot of money.’ Though controversy remains over the site’s legacy, clean-up was completed by 2005, costing $7 billion.
5f in use?
Meanwhile, Meyer’s group has been able to bind, split, activate and transform carbon dioxide in reactive coordination complexes with uranium in oxidation states from +3 to +5. His group has used such systems to insert carbon dioxide into bonds between uranium and ligands, convert it into isocyanates through multiple bond metathesis,5 and reductively disproportionate it to yield carbon monoxide and carbonate.6 ‘That led to a catalytic cycle where you can run a few cycles turning carbon dioxide into carbon monoxide,’ Meyer adds. ‘If you could optimise this cycle and feed carbon monoxide into a Fischer·Tropsch procedure, then you could make liquid fuels.’ These reactions shed light on uranium’s reactivity, he adds. ‘It has taught us that the bond in uranium chemistry is predominantly ionic, with some degree of covalency. That means the uranium·ligand bond is highly polarised, and therefore much more reactive than a wholly covalent bond.’
Mazzanti and her colleagues have also been investigating pentavalent uranium, having been the first group to probe the reactivity of pentavalent uranyl ions.7 But they’ve also been exploring another surprising new direction · using uranium to make single molecule magnets that can switch between magnetic states arising from spin alignments. Such molecules could be used to store information, for example as superior magnetic thin films to coat hard disks. They need high ‘magnetic anisotropy’, creating a barrier to switching, and high spin. Transition metals have high spin and actinides have high magnetic anisotropy.
‘There has been an idea in the community for a long time that uranium could establish magnetic communication with transition metals,’ Mazzanti says. ‘One problem in verifying this was building compounds that contained both actinides and transition metals. We built either uranium clusters or polynuclear compounds of pentavalent uranyl, which we associated with manganese. We demonstrated magnetic communication between them, which leads to single molecule magnet properties.8 It’s very important and will lead to future developments in the field.’
With Mazzanti looking beyond nuclear applications, catalysis and data storage are perhaps the most obvious practical use for uranium research. She admits that a general fear of the element is ‘a major problem’, though she echoes Liddle’s position that depleted uranium is no more dangerous than other heavy metals. But she emphasises that the possible benefits of studying actinides are also more profound. ‘We are doing fundamental research, which I think will lead to real applications, Mazzanti says. ‘But it also brings a new point of view for transition metals that could lead to important discoveries.’
Andy Extance is a science writer based in Exeter, UK
References
1 D L Clark et al, Inorg. Chem., 1989, 28, 1771 (DOI: 10.1021/ic00309a004)
2 I Castro-Rodriguez et al, Science, 2004, 305, 1757 (DOI: 10.1126/science.1102602)
3 D M King et al, Science, 2012, 337, 717 (DOI: 10.1126/science.1223488)
4 P L Arnold et al, Nature, 2008, 451, 315 (DOI: 10.1038/nature06467)
5 I Castro-Rodriguez, H Nakai and K Meyer, Angew. Chem. Int. Ed., 2006, 45, 2389 (DOI: 10.1002/anie.200501667)
6 O P Lam et al, Chem. Commun., 2010, 46, 3137, (DOI: 10.1039/b927142b)
7 F Burdet, J Pecaut and M Mazzanti, J. Am. Chem. Soc., 2006, 128, 16512 (DOI: 10.1021/ja067445t)
8 V Mougel et al, Nat. Chem., 2012, 4, 1011 (DOI: 10.1038/nchem.1494)
![Structure of [UCl4(HCN)4]](https://d2cbg94ubxgsnp.cloudfront.net/Pictures/380x253/4/5/2/135452_The-structure-of-the--UCl4-HCN-4--molecule-in-its-solid-state-compound.jpg)











No comments yet