Single atom and hierarchical nanopore catalysts are reducing the need for precious metals, and could clean up the energy and chemical industries, finds Andy Extance
Seemingly invisible entities catalysed a dramatic shift in Maria Flytzani-Stephanopoulos’ research. Her team at Tufts University in Massachusetts, US, was studying gold and platinum catalysts supported on ceria particles for the water–gas shift reaction. This process converts carbon monoxide and water to carbon dioxide and, most importantly, hydrogen. It’s a valuable source of hydrogen both for ammonia fertilisers and clean energy sources. Flytzani-Stephanopoulos and her colleagues tried to leach away all the metal using a cyanide-based process. Yet, as the team published in 2003, the remaining support was still just as active as a catalyst.1
‘A very small amount of residual gold or platinum strongly associated with ceria,’ Flytzani-Stephanopoulos recalls. ‘Now we know they were single atoms. This was so exciting that it made us go after atomically dispersed supported metal catalysts in full steam! The field is now booming, with more than 350–400 publications per year.’
Consequently, today chemists’ understanding of exactly how catalysts work is evolving to consider new factors. The focus of researchers like Flytzani-Stephanopoulos is zooming in to individual catalyst atoms. But at the same time such systems need to get reactant chemicals to those places. In some cases, this requires new structural solutions to problems at larger scales, also found with more conventional catalysts. And in exploring these new frontiers, researchers are making vital progress towards making our chemical and energy industries more sustainable.
Yet studying single atoms was right at science’s cutting edge when Flytzani-Stephanopoulos’ team found these compelling clues of their catalytic capabilities. ‘At that point, there was no real analytical technique available commonly to researchers that could visualise a single atom,’ explains Sharon Mitchell from the Swiss Federal Institute of Technology, ETH Zurich. ‘There were a lot of hypotheses but we couldn’t get a good fundamental understanding of these materials.’
A decade later, Mitchell’s colleagues in the ETH Zurich advanced catalysis engineering team, led by Javier Pérez-Ramírez, had a similar experience. The team sought improved catalysts for semi-hydrogenation of alkynes, for example converting 1‐hexyne to 1-hexene. Pérez-Ramírez received promising palladium-based materials from Markus Antonietti’s team at the Max Planck Institute of Colloids and Interfaces in Mainz, Germany.2 According to Mitchell, they expected palladium nanoparticles, ‘but we were initially unable to see the metal’. However, aberration‐corrected scanning transmission electron microscopy techniques developed in the 2000s ultimately enabled Pérez-Ramírez’s team to find that the palladium was present as single atoms. ‘It was very selective performance, which led us to wonder, what is the scope of these materials?’ explains Mitchell.

By this time, Tao Zhang from the Dalian Institute of Chemical Physics in China and colleagues were already exploiting extreme metal dispersion. Dispersion is important as many expensive metal atoms are trapped inside bulk catalysts and nanoparticles, and unable to participate in reactions. ‘Logically, by depositing metal atoms one by one onto a support we can achieve 100% utilisation of metals,’ Zhang tells Chemistry World.
Strong and stable
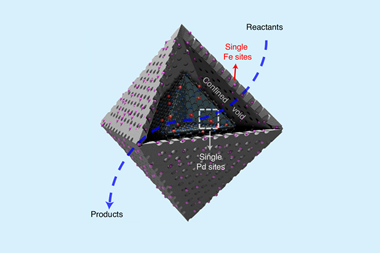
In 2009, Zhang and his team therefore co-precipitated single platinum atoms on to the surfaces of iron oxide nanocrystallites.3 They studied them with the new electron microscopy techniques, as well as spectroscopic and computational methods. The Dalian scientists showed that bonding between the oxide and platinum made the catalysts ‘highly stable and active for carbon monoxide oxidation’, Zhang says, and revealed the catalytic mechanism. ‘We were the first to prepare an unambiguous single-atom catalyst by a practical method, and to systemically characterise and identify the relationship between the catalytic performance and the single-atom catalyst,’ Zhang adds. Dispersing catalyst atoms at this extreme level also brings new chemical capabilities. Perhaps the most important are selective transformations, like the hydrogenation of functionalised nitroarenes, for example from 3-nitrostyrene to 3-aminostyrene, published by Zhang’s team in 2014.4
Pérez-Ramírez, Mitchell and their colleagues likewise further explored single atom catalysts’ capabilities. For example, they developed a palladium single-atom catalyst supported on carbon nitride for the Suzuki coupling reaction.5 ‘We showed through simulations and microscopy that we have a macroscopic carbon heterocycle with six nitrogens in it that can stabilise the palladium atom,’ Mitchell says. ‘This yielded very high stability, and a much higher turnover frequency.’ Furthermore, a solid catalyst can be reused easily, unlike the dissolved palladium catalyst typical for Suzuki reactions, lowering costs significantly.
The community has made substantial progress in preventing aggregation of single atoms
Yet there are relatively few examples to date of where single atoms can enhance catalysis in this way. ‘I think the scope has not been fully explored yet,’ says Mitchell. ‘We actually need to revisit a lot of applications and then maybe start the optimisation for the single atom and not for the established catalyst.’
Nevertheless, Flytzani-Stephanopoulos is surprised by how many reactions have already been found to be catalysed by supported single metal atoms. Her team’s explorations have included gold atom oxidation catalysts on various supports for important reactions including water–gas shift, methanol and ethanol dehydrogenation, and steam reforming of methanol.
An inherent challenge with single atom catalysts is that the atoms aggregate during fabrication, storage and catalytic reactions. However, Flytzani-Stephanopoulos says that there are now several approaches to preventing these problems. ‘Dynamic changes during reaction are often reversible in the right gas atmosphere,’ she says.
‘Although it is currently still a challenge to develop thermally stable catalysts, the community has made substantial progress in preventing aggregation of single atoms,’ adds Zhang. He highlights three prominent prevention strategies. One is introducing coordination atoms to bond to the single atoms strongly. For example, single atom catalysts can be secured on nitrogen-doped carbon supports, as in the ETH Zurich team’s Suzuki catalyst system. The second relies on very strong metal–support interactions, as in platinum–ceria and platinum–iron oxide systems. The third engineers defects in two-dimensional materials or oxides to support single atoms.
Recruiting alloys
A further option is delivering stable single atom catalysts in a different form altogether. Together with her Tufts colleague Charlie Sykes, Flytzani-Stephanopoulos received the 2019 American Chemical Society Catalysis Lectureship for their work on single atom alloys (SAAs), where a single metal atom is supported by another metal. She calls them ‘very promising for future applications’, particularly selective hydrogenation and dehydrogenation reactions. Flytzani-Stephanopoulos cites the example of selective dehydrogenation of butane to butene at 400°C on a platinum–copper SAA.6 ‘Here the support is metallic, a host, like copper,’ she explains, with single catalyst metal atoms doped into their surfaces only. ‘The result is a catalyst with better activity than the host and a better selectivity than the dopant metal, as good as that of the host. They also show good stability, tolerance to poisons like carbon monoxide and no propensity to carbon formation up to high temperatures.’

Flytzani-Stephanopoulos also works with the Integrated Mesoscale Architectures for Sustainable Catalysis (IMASC) Energy Frontier Research Centre. Funded by the Department of Energy and headquartered at Harvard, in IMASC the Tufts team studies nanoporous dilute alloys for selective hydrogenation and oxidation reaction systems.
Juergen Biener from the US Lawrence Livermore National Laboratory (LLNL) is also part of the IMASC centre. He agrees that nanoporous alloys can be very stable, but under some circumstances gases and liquids can only travel slowly through them, diminishing their benefits. Researchers therefore want to mimic ‘hierarchical’ nanoporous structures. Biener compares the widest pores to freeways, but notes that current production methods would create a nonsensical road system. ‘Either you are not allowed to put any freeways in the system, or they’re just randomly placed without planning. That’s the structure you get with traditional synthetic approaches to porous materials. In nature, the lung, your kidneys, liver, they all depend on very effective mass transport. Typically that involves hierarchical pore systems.’
Therefore, Biener and his LLNL colleagues have 3D printed such structures using gold and silver based inks.7 Their starting structure was similar to a woodpile, with solid sections stacked with micrometre-scale ‘freeway’ channels between them. The scientists then etched away, or dealloyed, the silver with nitric acid to create nanoscale pores, and used the resulting catalyst to oxidise methanol. ‘Pre-Columbian cultures used something similar to make metal objects to appear made out of pure gold whereas they were made out of copper–gold alloy,’ Biener explains. ‘It’s commonly used to create nanoporosity, but the resulting structure has poor mass transport properties.’ In LLNL’s digitally-designed catalyst, the remaining 2 atom% of silver enables the oxidation reaction, making this material a hierarchically structured SAA catalyst.

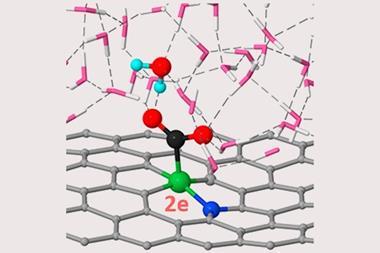
LLNL researchers are also working on using SAAs for electrochemical reduction of captured carbon dioxide to produce fuel or industrial feedstock chemicals. They are collaborating with global oil and gas company Total American Services in order to reach the scales needed. ‘Whether that technology will ever go online at a gigaton scale will depend on how efficient it is,’ Biener says. ‘There is hope that optimising the morphology and reactivity of the catalyst will contribute.’ This could involve relatively cheap, copper-based hierarchical porous SAA. ‘The stability of SAAs is an amazing finding because nanoparticle-based catalysts are prone to deactivation due to coarsening and agglomeration,’ Biener adds. ‘Specifically with gold catalysts, this is a really big problem because experience shows that only nanoparticles below 5nm have catalytic activity. Our materials started out at 30nm and they don’t lose their activity.’
Marc-Olivier Coppens from University College London in the UK works on designing nanoporous materials, using ‘nature-inspired engineering’. His team ‘exploits nano-confinement effects as well as optimised transport across length scales, guided by nature’. ‘In catalysis, we’d like to have a very high specific surface area,’ he says. ‘But as a result of this, typically the pores are very narrow, so you get transport limitations. As a result, we’d like large pores to have transport towards the small pores, and for molecules to easily diffuse in and out of the porous catalyst.’
Take your breath away
Strikingly, his group is applying the concept to hydrogen fuel cells that generate electricity.8 ‘Even though fuel cells have been around for 100 years, the catalysts are often noble metal based, so they’re expensive, and there are scalability and stability issues,’ he says. ‘We’ve redesigned the fuel cell, thinking about how the lungs operate. The lung has a very intricate hierarchical structure. Air flows down by pressure-driven flow, until it reaches your bronchioles. Then diffusion sets in, so it’s no longer flow-driven, it’s diffusion-limited. At the transition, the structure changes from fractal self-similar to uniform. In addition, the sequence of channels is optimally proportioned to lead to minimum metabolic energy losses.’
Using this concept in fuel cells ‘you can distribute oxygen and fuel optimally, first by flow, and then by diffusion down to the active sites,’ Coppens says. ‘The surprise in this is that you are able to reduce the amount of required metal by an order of magnitude.’ The team is now exploiting hybrid fuel cell–battery designs in a mobility scooter on the UCL campus. ‘That tells you that we are approaching meaningful applications, which is quite exciting.’
Can we design a catalyst structured like a lung?
Yi Cui from Stanford University in California, US, is likewise exploring both single atom catalysis and optimising mass transport. ‘Mass transport’s significance is more than people expected previously,’ he tells Chemistry World. ‘It can play a huge role to help reducing costs.’ His team is addressing what Cui calls ‘one of the big problems in catalysis’. ‘Many important reactions involve gas molecules, which need to get in contact with catalysts, coming into a water environment,’ Cui says. ‘To get the reaction to happen effectively, you need three phases in contact.’ But as the gases are not very soluble in water, very little diffuses to the catalyst. So Cui had an idea. ‘Can we design a structure that could allow us to transport our gas molecule directly to solid catalysts, without going through liquid, but at the same time have solid catalysts in contact with the liquid?’
Cui and his colleagues – including former US Secretary of Energy Steven Chu, now at Stanford – also look to the alveolus structure in mammalian lungs. ‘When we inhale air, it comes all the way through the lung, to the end alveolus,’ Cui says. ‘Basically it’s a gas bubble, and then there’s the membrane of the alveolus. Gas goes through the membrane, into the blood right away. The gas molecule never really needs to be soluble in the liquid in a significant way before it reaches the red blood cells. We said “Can we design a catalyst like this?”’
They have developed gold–polyethylene membranes with high gas permeability that allow water through reluctantly. This allows them to bring together the gold catalysts with both liquid and gas phases at the same time. The Stanford researchers have used the technique both to electrochemically reduce carbon dioxide to carbon monoxide,9 and in hydrogen fuel cells.10
The team’s catalysts use less expensive precious metal, ‘yet still maintain a high production rate, because we are transporting the gas molecule in and out so fast’, Cui says. ‘In the past, people might increase the loading because their mass transport is not efficient.’ The Stanford team is now exploiting the approach in an industry-funded project. ‘We are building integrated three dimensional devices, a kind of flow system,’ Cui says.
Innovations such as single-atom catalysts and hierarchical nanoporous structures are pushing the limits of what chemistry can achieve even further than ever before. The collaborations and prototypes exploiting them are also early signs that these innovations will enter the mainstream. In revealing what was invisible, and learning from nature, researchers are enabling chemists to address issues of sustainability that challenge the entire world. And these are just initial steps, Cui feels. ‘I expect that down the road, there will be more examples that might present themselves in a different morphology,’ he says.
Andy Extance is a science writer based in Exeter, UK
References
1 Q Fu et al, Science, 2003, 301, 935 (DOI: 10.1126/science.1085721)
2 G Vilé et al, Angew. Chem. Int. Ed., 2015, 54, 11265 (DOI: 10.1002/anie.201505073)
3 B Qiao et al, Nat. Chem., 2011, 3, 634 (DOI: 10.1038/nchem.1095)
4 H Wei et al, Nat. Commun., 2014, 5, 5634 (DOI: 10.1038/ncomms6634)
5 Z Chen et al, Nat. Nanotechnol., 2018, 13, 702 (DOI: 10.1038/s41565-018-0167-2)
6 G Giannakakis et al, Acc. Chem. Res., 2019, 52, 237 (DOI: 10.1021/acs.accounts.8b00490)
7 C Zhu et al, Sci. Adv., 2018, 4, eaas9459 (DOI: 10.1126/sciadv.aas9459)
8 P Trogadas et al, Energy Environ. Sci., 2018, 11, 136 (DOI: 10.1039/c7ee02161e)
9 J Li et al, Nat. Catal., 2018, 1, 592 (DOI: 10.1038/s41929-018-0108-3)
10 J Li et al, Joule, 2019, 3, 557 (DOI: 10.1016/j.joule.2018.11.015)










No comments yet