Elinor Hughes catches up with some of the latest materials and processes in photovoltaics research
Solar energy is the most abundant energy resource on Earth, and solar panels can convert this energy into electricity directly. But the photovoltaic cells that make up solar panels currently produce energy more expensively than burning fossil fuels. So there’s a big drive to bring down the material and processing costs, and improve their efficiency, while addressing the growing need for renewable energy sources.
The majority of photovoltaic (PV) cells on the market are silicon-based. Silicon represents a first generation PV technology, which is costly to produce but efficient. The second generation consists of thin-film PV, such as the inorganic materials cadmium telluride and Cigs (copper indium gallium diselenide, CuIn1–xGaxSe2), and these are cheaper to produce. Organic, perovskite and dye-sensitised solar cells are usually referred to as third generation – and these have the potential to further lower cost, says Aron Walsh from the University of Bath, UK.
Perovskite push
Walsh works on next generation solar cells that are being developed to achieve a high performance at a low cost, while eradicating the need for low-abundance or toxic elements. ‘We’ve taken inspiration from materials that occur naturally as minerals – we call this solar mineralogy,’ he says. Some of these new materials include kesterites – sulfide minerals with the formula Cu2(Zn,Fe)SnS4, which are being developed to enter the thin-film market. The UK consortium PVTEAM, of which Walsh is a member, is working on a modification with copper tin zinc sulfide. Copper, zinc, and tin are abundant, and kesterites are as good as cadmium telluride and Cigs in terms of physical properties, says Walsh.

Walsh also works with perovskites – ABX3 minerals with the same crystal structure as calcium titanate (CaTiO3). The materials are Earth-abundant and the simple solution processing required suggests that manufacture at scale should be possible, says Walsh.
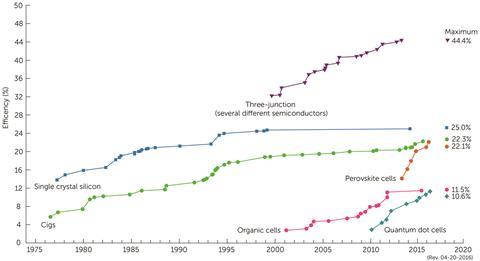
In 2009, organic–inorganic perovskites were initially used in dye-sensitised solar cells to absorb light as a replacement for the molecular dyes. Then, in 2012, Henry Snaith and colleagues at the University of Oxford, UK, found that the perovskite itself was capable of acting as the photoactive semiconductor. Perovskites have since seen a boom in popularity in PV research. They are cheap, easy to make and can be made thin and flexible enough to manufacture in a roll-to-roll process. They can also be layered on conventional silicon solar cells to boost their energy output. The best devices so far have reached conversion efficiencies – a measure of how much of the sun’s energy is converted into electricity – of over 22%. Crystalline silicon PV on the market have conversion efficiencies of up to 23%, but it’s difficult to compare the two because the result for silicon is for a 1m2 module, while the record perovskite efficiencies are for 0.1cm2 test cells, says Walsh.
Several groups are working on making perovskite devices less toxic (typical devices contain lead), improving their stability (one theory is that iodine-rich regions form in the cell under light, restricting the voltages obtainable in the device) and making them less sensitive to wet weather and humid conditions. Snaith and his team have recently improved the moisture resistance in metal halide perovskite solar cells. They discovered that a commonly used hole-transporter dopant (hygroscopic lithium salt) makes the top layer of the cells hydrophilic, causing them to degrade in the presence of moisture. So they used low cost, hydrophobic hole transporters in conjunction with a doping method incorporating a pre-oxidised salt of the respective hole transporters to prepare efficient perovskite cells with greatly enhanced water resistance.
Walsh and his team are still searching for other next generation candidate inorganic materials. They have just completed a computer screening of over four trillion compounds and are in the process of analysing the data.
Material gains for organics
Physicist Moritz Riede from the University of Oxford in the UK is working towards enhancing the efficiency and lifetime of organic semiconductor photovoltaics. These materials have already undergone large-scale commercialisation for use in organic light emitting diodes for mobile phone and television displays. Riede’s team is using the same processing method – vacuum deposition of purified small organic semiconducting molecules – for their cells. The technique allows layers to be built up under controlled conditions until a working organic solar cell is produced.
Organic solar cells have several advantages over existing solar cells. They are very thin and lightweight, can be bent and rolled up, are robust, come in different colours and can even be made semi-transparent to make electricity-generating windows, explains Riede. They are also based on abundant and non-toxic raw materials and the manufacturing techniques – printing or vacuum coating – are well suited for coating large areas rapidly at low cost and with a low energy input.
The question is, can we better understand the physics behind efficiency losses and find ways to get more efficient organic solar cells?
Moritz Riede
Unfortunately, at the moment, they are more expensive to make than commercially available solar cells, given that they have so far only been produced on a small scale using custom-made equipment. ‘The economies of scale that helped bring down the cost of silicon solar cells significantly over the last decades have not kicked in yet,’ says Riede. ‘I see the potential cost for electricity generated by organic solar cells, when produced on large scale, below that of silicon solar cells.’
But how good are they at converting solar energy into electricity? The certified laboratory efficiency record for organic solar cells is currently 13.2%, which does not seem high compared with the overall world record of 46%, says Riede, but he is keen to point out that efficiency alone as a measure is not sufficient. ‘What matters in the end is how much the solar electricity costs, and that is one reason why silicon solar cells with an efficiency of around 20% are currently the commercially most successful solar technology,’ he says.
The standard way to compare different technologies is at full sunlight with the solar cell temperature at 25°C, but this does not capture how much energy is harvested over the course of time, with varying light intensities and solar temperatures that are usually much higher than 25°C (after all, you put solar cells in the sun), says Riede. ‘Under real-life conditions, over the course of one day, organic solar cells perform better, such that their 13% conversion efficiency probably compares better to a 15% certified silicon solar cell.’
Riede sees organic solar cells being used first in markets in which conventional solar cells struggle; for example being integrated into buildings, on cars, in windows, on mobile electronic devices, on marquees and tents, as well as eventually rooftops and power stations. His next step is to better understand what currently limits the conversion efficiencies of organic solar cells. ‘There are some ideas on what causes losses in efficiency and how to reduce them. The question is, can we better understand the physics behind those losses and find ways to actually reduce them to get more efficient organic solar cells? I think research will remain interesting for many years to come,’ he concludes.
Improving thin-film processing
Two UK companies that are developing more efficient processing techniques are Nanoco Technologies and Eight19. Nanoco, based in Manchester, is a world leader in the manufacture of cadmium-free quantum dots and other nanoparticles. Its head of solar, Stephen Whitelegg, is in charge of device engineers and chemists who work on developing solution processes to produce thin-film inorganic semiconductor Cigs. This is currently produced by sputter coating or evaporation in a vacuum. But these techniques are expensive and the throughput can be slow, says Whitelegg, who adds that because of the cost, Cigs only have a 5% share in the photovoltaics market.
Cigs outperform silicon in terms of energy produced over the life cycle of a solar panel (the levelised cost of electricity) because they are more efficient at converting the sun’s energy in low light conditions and they’re better in hot environments. With Cigs, the band-gap (the energy difference between the top of the valence band and the bottom of the conduction band) can be tuned to gather as much of the solar spectrum as possible by altering the ratio of gallium and indium; this tuning can’t be done with silicon. Cigs modules can also be more aesthetically pleasing with their uniform black appearance, instead of the individual wafers held together by wires that are prominent in silicon solar cell modules. The cells can be integrated into other materials, such as roof tiles, and fabricated on flexible substrates making them suitable for the building integrated PV market, says Whitelegg.
Whitelegg’s main aim is to reduce the cost of producing Cigs, while maintaining a high performance. Their process involves coating a rigid substrate, such as glass, with an ink made up of inorganic semiconductor nanoparticles. ‘Printing is much cheaper than using a vacuum technique and with vacuum, at least 30% of the material is wasted, whereas printing produces less than 5% waste,’ says Whitelegg. The semiconductor nanoparticles are stabilised by organic ligands and dispersed in an organic solvent to form an ink. Once the ink is printed on the substrate, the layer is heated to remove the ligands and to sinter the nanoparticles together to produce a thin film. By growing the nanoparticles in this bottom-up approach, the team is able to get particles just 3nm in size, unlike the top-down approach, in which the full material is ground up in a ball mill to produce 50nm particles. Smaller particles pack more closely together in large amounts on the substrate and there is a better stoichiometric uniformity (the ratio of elements within the particles), both of which lead to better performance.
In terms of conversion, the team’s Cigs have reached 16.7% (active area). The best conversion efficiency achieved so far in the lab with vacuum techniques is 22.1%, but Whitelegg points out that scaling up to module will cause it to drop to 14–16%. ‘This is where solution processing for Cigs has the potential to achieve a higher module efficiency due to improved stoichiometric uniformity,’ he says. ‘We are targeting at least 20% on the R&D scale before we start commercialising.’ His team is now looking forward to scaling up and commercialising their product.
Cambridge-based Eight19 – which takes its name from the time it takes sunlight to reach the earth (eight minutes and 19 seconds) – is a company that is developing new low-cost, flexible plastic solar cells. Jurjen Winkel heads the architecture development team that evaluates materials, and establishes and tests processes required to make solution-processed solar cells scalable for roll-to-roll production.
‘The cells are based on a mixture of non-toxic organic and inorganic materials that can be processed into inks, which are deposited in layers onto flexible supports to create serially-interconnected photovoltaic modules,’ explains Winkel. The reason they use solution-processed organic photovoltaic modules is because they are processable at temperatures below 150°C, enabling the use of low-cost plastic supports required for roll-to-roll processing. Roll-based processing is normally used as a high speed technique to produce osmotic membranes and holographic films, for example, he says.
With flexible solar cells, more variety can be introduced into their design, such as varying their shape and colour to meet customer requirements. ‘Unusual shapes and configurations enable design aspects of organic photovoltaic (OPV) materials to come to the fore,’ says Winkel. ‘The solar modules can be made to look like tinted windows, allowing unconverted light through the entire stack. This feature makes OPV interesting for building integrated solutions and allows considerable architectural creativity.’

At Eight19, the best cells have a conversion efficiency of around 7.5% using an entirely scalable process, but they are working on improving this figure, as OPV cells have been known to reach efficiencies in excess of 10%. ‘It is important to understand that these are typically achieved using very small test cells fabricated on glass using unscalable techniques such as spin-coating and with pretty near unlimited processing time windows,’ says Winkel. ‘The challenge of maintaining high efficiency when transferring to plastic substrates at scale and in a manufacturing process where there is a limited time for each step should not be underestimated.’
As well as gathering the sun’s energy, OPV cells can also be used to harvest energy from artificial light. Artificial lights emit mainly in the visible region of the electromagnetic spectrum, which OPV cells absorb well. ‘As a result, OPVs have significantly higher efficiency than other types of cells when it comes to artificial lighting, especially LEDs, as the bulk of the emitted light can be absorbed,’ says Winkel, who is currently running tests on OPV cells in this area.
Eight19’s focus over the next two years is to commercialise their products. ‘The challenges the company faces are the same as for any young company – achieving sufficient scale to realise the significant cost potential of OPV materials and to ensure our offerings match the expectations of first mover customers. Finding a niche market where OPV excels is key to bridging the scale gap,’ says Winkel.
As well as reducing cost, the biggest challenge facing PV researchers is scaling up new materials and processes to work well in commercial devices. ‘Scale-up, while maintaining high performance, can be just as demanding as identifying a viable photoactive material,’ concludes Walsh.
Elinor Hughes is a science writer based in Bridgend, UK











No comments yet