From mummification to metallurgy, Rachel Brazil looks at the impressive chemistry used by this ancient civilisation
It’s a busy year for Egyptologists; 2022 marks the centenary of the discovery of the tomb of King Tutankhamun in the Valley of the Kings by British archaeologist Howard Carter. Stephen Buckley, an archaeological chemist and expert in mummification at the University of York, UK, says he has just returned from a symposium at Highclere Castle, organised by Lady Carnarvon, the great granddaughter-in-law of the fifth Earl of Carnarvon, who funded Carter’s expedition back in the 1920s. The exquisite artefacts excavated from the 3350-year-old tomb have continued to inspire a sense of awe in those who visit them – and raise questions as to the technical skills and scientific abilities of the ancient Egyptian creators.
Of all the sciences, chemistry is the one with which ancient Egyptians are most closely associated. One school of thought even attributes the word chemistry to the Egyptian name for Egypt, handed down via Greek and Arab alchemists. The word kemet means ‘black land’, describing the colour of the fertile black soil annually deposited from the Nile. ‘It’s no accident that this was the name [and] a recognition that the Egyptians were the best chemists of the ancient world… even the Greeks reluctantly admitted [it],’ says Buckley. But Sydney Aufrere, an Egyptologist from the University of Provence-Aix-Marseille in France, says that although it seems logical, ‘there is nothing conclusive in this hypothesis’.
It’s certainly true that the ancient Egyptians became skilled in what Marco Beretta, a historian at the University of Bologna in Italy, calls ‘the chemical arts’. Beretta has recently edited a comprehensive account of the cultural history of chemistry in antiquity, which sets out how highly developed ancient civilisations used and controlled chemical knowledge. While practical skills existed, Aufere defines the Egyptian use of chemistry as ‘mytho-scientific’. They believed, for example, that materials possessed essences or emanations of different gods – magnetite was associated with the god Horus, natron salt with Osiris and tree resins were thought to be the tears of gods.
The use of chemistry seems widespread, but its connection to religious ritual was central, with metallurgists, perfumers and dyers working just outside temples, according to ritual rules. This led to secrecy surrounding their chemical knowledge. ‘Texts that remain of a technical nature, without mythological additions, are rare,’ says Aufere. ‘However, the ideas that we find exposed in the temples most probably emerged in trades that were able to observe the production of phenomena of a physical–chemical nature.’
Colourful chemists
Without texts, what’s left is the material evidence, says Beretta: ‘This huge array of items that shows chemical skills of the Egyptians.’ But what we don’t know is whether their achievements were more than just empirical trial and error – did they grasp any basic chemical principles? Aufere says it’s unlikely they were capable of rationally explaining the phenomena they observed outside their religious framework, but chemist Jehane Ragai, now an emeritus professor at the American University in Cairo, Egypt, isn’t so sure. On studying some of their achievements, she was surprised to find ‘there is some evidence that their chemical knowledge was maybe more than we are led to believe’.

At the core of the chemists’ skill set is synthesis, and the ancient Egyptians were the first civilisation to synthesize an inorganic pigment – Egyptian blue. Beretta says this was part of their wish to imitate the precious stones they associated with the gods and particularly lapis lazuli, the rare lazurite-containing blue aluminium silicate mineral, which they believed formed the god’s hair. From 2500BCE they began to synthesize the blue pigment calcium copper tetrasilicate (CaO.CuO.4SiO2). It was manufactured into the Roman era, before the technology was lost, only being rediscovered in the 19th century.
Ragai says looking at the chemistry involved you can’t help but appreciate their skill. ‘One of my students tried to prepare some Egyptian Blue, and you have to have an oxidizing atmosphere, and a catalyst.’ The raw materials – silica, lime and copper – would need to be heated with a flux such as borax to obtain the blue colour. ‘The stoichiometry of the end product had to be respected also. So again, here, we do have some chemistry,’ says Ragai. There is also evidence that the Egyptian chemists were able to tweak their furnace conditions and cooling rate to deliberately create an alternative green pigment.
The ancient Egyptians couldn’t have been aware of the recently observed fluorescence of Egyptian blue in the near-infrared region – which has become a simple way for conservators to identify its presence on artefacts and may also suggest applications in modern technologies, including most recently for battery electrodes.
Chemical make-up
One of the most important pieces of the jigsaw of ancient Egyptian chemistry is the discovery of synthetic lead pigments used in the kohl eye make-up worn by ancient Egyptians, dating back to 2000BCE. Published in 1999, chemical analyses carried out by Philippe Walter at the Louvre Museum in Paris, France, showed that the well preserved compounds contained in the Louvre’s collection of ancient Egyptian make-up pots included natural pigments galena (PbS) and cerussite (PbCO3), but also laurionite (Pb(OH)Cl) and the white powder phosgenite (Pb2Cl2CO3). Both are rarely found naturally and are unlikely to be the decomposition products of anything that is. Walter concluded that these must have been synthetically made, using what we would today call wet chemistry.
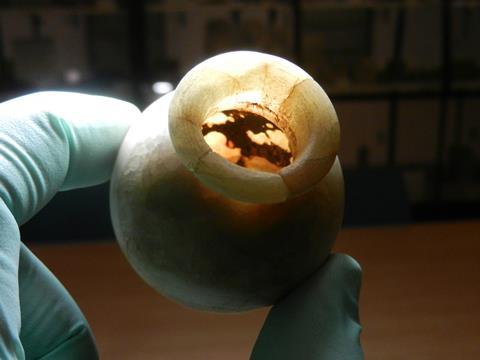
Based on later Roman recipes, Walter experimented with a method to produce phosgenite. He used the mineral litharge (PbO) crushed with salt and mixed in water and in some cases sodium carbonate. He neutralised the alkaline solution produced (the Roman recipe describes decanting off the solution and replacing the water each day to control the pH) and after several weeks identified either a laurionite or phosgenite precipitate, with crystals of similar size and morphology to those found in the Egyptian samples. Their capability to carry out a relatively complex reaction still surprises Ragai. ‘In this particular instance, [this] indicates that they might have had some knowledge of pH control, buffering action and of some basic quantitative chemistry.’
Walter’s work has since been confirmed by several research groups. In 2018, Lucile Beck, from the carbon 14 measurement laboratory at the University of Paris–Saclay, used radiocarbon dating to prove ancient Egyptian make-up samples included synthetic compounds. Carbonates from geological sources are too old to still contain C-14, but those created with atmospherically derived carbon (such as carbonate salts from evaporates) in the last 3000 years would be datable. Beck tested cosmetics from the Louvre collection, including some from the famous 18th century expedition of Napoleon Bonaparte, which also yielded the Rosetta stone. Finding such samples isn’t easy, she adds – in the past archaeologists were only concerned with artefacts and frustratingly often cleaned out any materials inside.
From the samples tested, one was confirmed as a synthetic cosmetic, made from the same phosgenite compound discovered by Walter and dated from 1763–1216BCE. Beck was also able to carbon-date the wooden pot (to 1514–1412BCE, during the reign of Amenhotep III) that contained the make-up. ‘We obtained a very good correlation between the wooden box results and the cosmetic results. So in that case, [it] really is direct proof that this cosmetic was artificially produced at that time,’ she concludes. This may be the oldest evidence for the use of wet chemistry to produce cosmetics, although given only one of the samples was synthetic, it seems rare. Beck has also tested some samples from the British Museum and so far has found no synthetic cosmetics.
It’s very likely that our current distinctions between cosmetic, medical, ritual and religious did not fit at the time of the ancient Egyptians
More evidence of synthetic phosgenite has come from a team from the research lab for archaeology at the University of Oxford in the UK, sampling 11 kohl containers in University College London’s Petrie Museum. They did a wide range of chemical analysis to pick up both inorganic and organic components. They also found lead compounds that are likely to have been synthesised. They found a much greater variety than previous modern analysis, ‘including things like minerals containing manganese [and] copper’, says Marabel Riesmeier, a chemist in the department of history and philosophy of science at the University of Cambridge UK, who worked on the project. She adds they also often found constituents that would not naturally occur together, so there was a deliberate intent in their formulations.
In 2010 a French team, including Walter, suggested that the kohl formulations made by the ancient Egyptians may have been formulated for their medical properties. At submicromolar concentrations they showed Pb2+ ions can elicit a specific oxidative stress response in skin cells that can stimulate an immune response. Given the toxicity of lead this seems counter-intuitive, but Riesmeier says the dangers are sometimes exaggerated. ‘Most of the salts that are in [the make-up] are not very soluble.’ Whether lead was selected for medicinal benefit is highly speculative, but Riesmeier concludes, ‘it’s very likely that our current distinctions between cosmetic, medical, ritual, [and] religious did not fit at the time of the ancient Egyptians’.
Marvellous mummies
Riesmeier and her colleagues also identified a wide range of organic substances of both animal and plant origins, and many reminiscent of organic materials associated with mummification – the embalming process now synonymous with the ancient Egyptians. Buckley has extensively studied mummification and the evolution of the chemical preparations developed. Although early mummification was initially judged to be simply desiccation caused by the hot, dry Egyptian desert, Buckley has unearthed evidence of embalming as early as the late neolithic pre-pharaonic era, over 6000 years ago. ‘At this time, they’re already using natural products, [tree] resins, plant-based products that have antibacterial, antifungal and insecticidal [properties] to some extent,’ says Buckley. ‘We’re not talking about excellent, full-body preservation at this time, but it would have had some effect on the preservation.’

By the pharaonic period, the ancient Egyptians had stepped up their mummification game, largely due to the use of natron, a mineral deposit consisting of hydrated sodium carbonate with additional sodium salts (bicarbonate, chloride and sulfate), found in Wadi Natrun in northern Egypt. Buckley suspects it was first used in solution to preserve organs around 2600BCE, once the Egyptians realised that removing them from the body would aid preservation. Buckley believes that this shows the Egyptians understood that natron was more than just a desiccant, but had more extensive preservative properties. At different historical periods, it was used to mummify bodies as a dry powder or in solution.
Tutankhamun was likely to have been mummified in a natron solution, according to Buckley. The Egyptians’ mummification skills were at their height during his reign in the 18th dynasty. The idea was mentioned in 1914 by chemist Alfred Lucas, the original conservator who worked with Carter and who was known as the Sherlock Holmes of Egyptology. His analysis of the mummy found spots of sodium chloride and sulfate on the skin, but no carbonates. This puzzled him and he eventually concluded that natron had not been used at all. Now Buckley says this would be characteristic of a solution. ‘You’ve got those two salts left that are just neutral salts [and] just sit there, but you don’t have the carbonate and bicarbonate because they’ve combined with the tissue.’
In solution, the high pH would ‘stop the bacteria in their tracks’, says Buckley, by preventing enzyme function. The solution can diffuse deeper into tissue and change the collagen structure, possibly by cationic binding to protein carboxyl residues, providing even more complete preservation. Buckley says the result would not be the ‘somewhat emaciated royal mummies that we see today’, which have become desiccated over time; ‘the body would still have a significant amount of tissue and water within it’. From his own mummification experiments, including on a donated human body, Buckley concludes that, when done extremely carefully, the result would have been almost perfect preservation. ‘As they go into the tomb, they look exactly the same [as when they were alive]; they look as though they’re asleep.’

Another discovery that Buckley says shows a high level of sophistication and control was their use of organic resins from a variety of sources. In addition to using these mixtures as waterproofing, evidence shows these were also applied to bodies before using natron. ‘Because of the caustic nature of the carbonate and bicarbonate, if you don’t apply this organic layer, it bleaches the skin ghost white. If you put on this organic barrier material first and then put it in a natron solution, what you then get is excellent preservation, but it also maintains the skin tone,’ explains Buckley.
But how much chemical knowledge these advanced funerary practices indicate is debatable. ‘Clearly, some of it was empirical, experiment and observation… but I suspect there was some detailed understanding of particular products being used in very particular circumstances,’ says Buckley. Ragai points out that some surviving ancient Egyptian medical texts, such as the Edwin Smith Papyrus, thought to have Old Kingdom origins, show a rational and almost scientific approach to diagnosis, much in keeping with what we would see as the scientific method. ‘If they had that in their medical approach, then the same reasoning could be applied to the chemical path,’ she suggests.
Skilled metalworkers
The ancient Egyptians also showed immense technical skill in metal working. They were never able to smelt iron – the only iron they had was meteoric and appropriately called the ‘metal of heaven’ – but they were masters at working copper, silver and gold. They produced objects such as Tutankhamun’s gold mask, made from copper-alloyed 23 karat gold, with quartz and obsidian eyes and inlaid lapis lazuli. One technique that impresses Ragai is their soldering method, known as colloidal or chemical hard soldering, seen in the fabrication of gold jewellery.

This used a powdered copper salt such as malachite (basic copper carbonate), glued in place. When heated together the salt is reduced and the melting point of the gold in contact decreases. The copper and gold alloy to form the weld. ‘Again, this is chemistry,’ says Ragai.
So would we recognise anything like a traditional chemistry lab in the workshops of ancient Egypt? Relatively little is known about their labs and equipment. A number of Egyptian temples have rooms that have been called laboratories. ‘Texts engraved in what is called the Laboratory [at the temple at] Edfu, a room where the recipes of perfumes and the list of olibanum [frankincense] are engraved… there are also recipes evoking the dyeing processes, notably at the temple of Dendara,’ says Aufere. In pharaonic times, there almost certainly wasn’t any of the glassware we might associate with a laboratory – glass blowing did not reach Egypt until the Roman empire.
Chemistry and chemical products were then, like now, really very important
The lack of chemical texts has also left us short on information on how the Egyptians viewed the physical world and if they had any theoretical framework to describe the nature of matter. This type of thinking is often attributed to the Greeks, such as Democritus who came up with the idea of the atom around 400BCE. Beretta says this focus on philosophical thinking may be due to the Greeks’ relative lack of natural resources and materials which prevented them from developing the practical and experiential skills of the Egyptians.
It is likely that much of the science that is now considered a product of Greek culture was handed down from Egypt. ‘All sorts of recipes, how to make artificial pearls or artificial precious stones… these are much older recipes that have been traced back [to the] Egyptian tradition,’ says Beretta.
Buckley agrees. ‘In terms of practical chemists, they were actually using some of the things we would recognize in the lab, long before Aristotle and others, [who] are the first to be credited with some of this experimentation.’
‘It shows that ideas and material and people circulated a lot more than we thought,’ says Beretta. ‘In the case of chemistry that is not surprising, because chemistry, chemical products and chemical commodities then, like now, were really very important.’
Rachel Brazil is a science writer based in London, UK













No comments yet