Scientists develop artificial muscle from proteins that mimic elastic and mechanical properties of natural muscle
Scientists in Canada and the US have developed artificial proteins that mimic the elastic and mechanical properties of the muscle protein, titin. When cross-linked into biomaterials, these proteins are tough and stretchy just like muscle tissue, the researchers say.
There has been intense research to develop synthetic elastomers that mimic muscle tissue for use in biomedical applications. However there are limitations in using these materials for implants as they cannot help with tissue repair or regeneration, and the artificial material can often be attacked by the immune system and rejected by the host’s body. The development of artificial muscle tissue using proteins could help overcome these problems and be used to replace damaged muscle tissue or in new prosthetics.
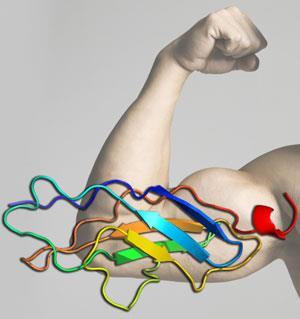
Titin, also known as connectin, is a muscle protein in humans, and is important in the contraction of striated muscle tissue - skeletal or cardiac muscle. Titin is the largest single polypeptide found in nature and is largely responsible for the passive elastic properties of muscle.
Hongbin Li and colleagues at the University of British Columbia in Vancouver and co-workers at Virginia Polytechnic Institute and State University have engineered biomaterials consisting of globular proteins - molecular springs derived from Streptococcus bacteria - and amino acid sequences derived from resilin, an elastomeric protein. They used genetically modified Escherichia coli bacteria to express the proteins and a photochemical reaction to cross-link the molecules, producing a gel-like material. The resulting elastic material recovers well at low strain and shows great toughness at high strain.
’Due to its unique mechanical and functional properties, this biomaterial may find potential applications in tissue engineering,’ says Li.
The material is ’highly resilient but also displays force damping properties when subject to high extensions,’ explains Elliot Chaikof, an expert in artificial muscle design at the Emory University School of Medicine, Atlanta, US. ’The principles established in this study provide an opportunity to expand the toolbox of molecular building blocks for fabricating new materials and devices for significant utility in medicine,’ he adds.
Li and his team are continuing to perfect and improve the material: ’We are currently developing robust methodologies to tune the mechanical properties of this biomaterial as well as designing new generations to incorporate functional and bioactive moieties,’ says Li.
’This is just the beginning of a very exciting new area of research, and many possibilities are ahead.’
Mike Brown
References
S Lv et al.Nature, 2010, DOI:10.1038/nature09024






No comments yet