Isolation of compounds from natural product mixtures simplified
US scientists have come up with a method that makes it easier to extract compounds that are difficult to isolate from crude natural product mixtures.
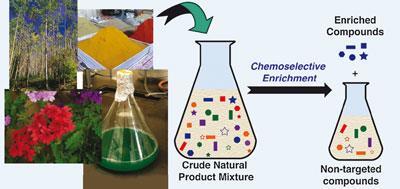
Erin Carlson and her team at Indiana University, Bloomington, used resins to target and isolate desired compounds in crude extract mixtures, in this case alcohols.
Isolating compounds from natural product mixtures is important because of their high propensity to interact with biological targets. Nearly half of currently available drugs are from natural products and pharmaceutical companies analyse crude extracts from, for example, plant materials for biological activity. Any active compounds are purified either by extraction and/or chromatography.
Current extraction methods rely on the compounds’ physical properties such as solubility, polarity or size. Despite advances in separation technology, purification is still problematic and time-consuming. ’Isolation of trace quantities of new natural products is often thwarted by the crude extract containing a plethora of compounds, with a heavy reliance on multiple liquid chromatography processes,’ says Gordon Florence, an expert in bioactive natural products from the University of St Andrews, UK.
Carlson’s team used a silyl-functionalised resin to capture and bind to the alcohol anisomycin - a protein biosynthesis inhibitor - from a mixture of compounds extracted from soil bacteria Streptomyces griseolus. The team washed the resin to remove non-targeted compounds before cleaving the anisomycin from the resin and found that the resin didn’t bind to compounds with functional groups other than alcohol. Following regeneration, the resin could be reused.
Florence believes that Carlson’s work ’will no doubt benefit the natural product community greatly in terms of both extracting and enriching components in minute quantities as part of the isolation and clean-up process.’
In the future, Carlson aims to build up a large toolkit of functional-group targeting tags. She believes that ’generating a large library of tags will provide an essential insight into the use of reversible, chemoselective reactions in other applications.’ These applications could include synthetic scavenging, isolation reagents and metabolomic profiling.
Mary Badcock
Link to journal article
Chemoselective enrichment for natural products discoveryAntoinette Y. Odendaal, Darci J. Trader and Erin E. Carlson,?Chem. Sci., 2011, 2, 760DOI:10.1039/c0sc00620c






No comments yet