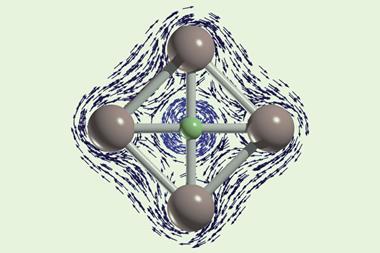
The first fluoronium [C–F–C]+ crystal ever grown has finally proven that it is a symmetrical structure – not a carbocation like some earlier research had suggested. Despite being the most electronegative element in the periodic table, fluorine engages in two almost covalent bonds with its neighbouring carbon atoms.
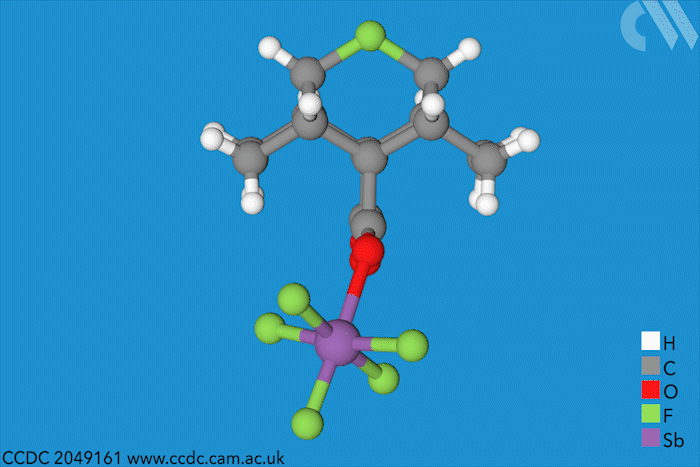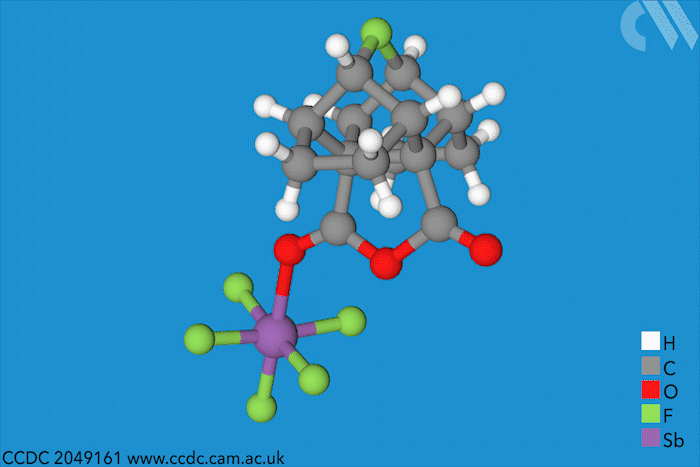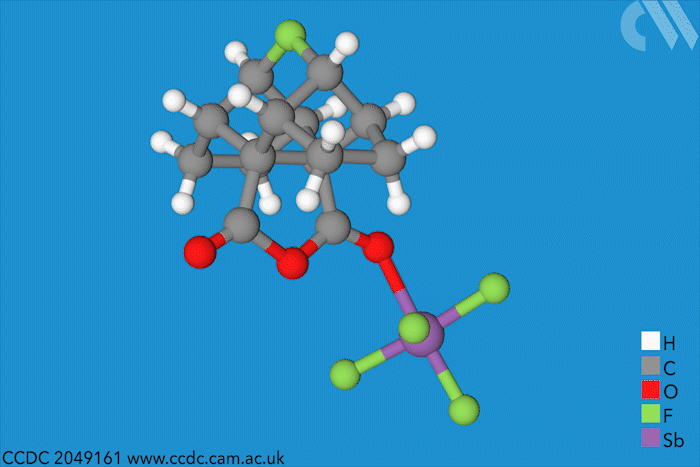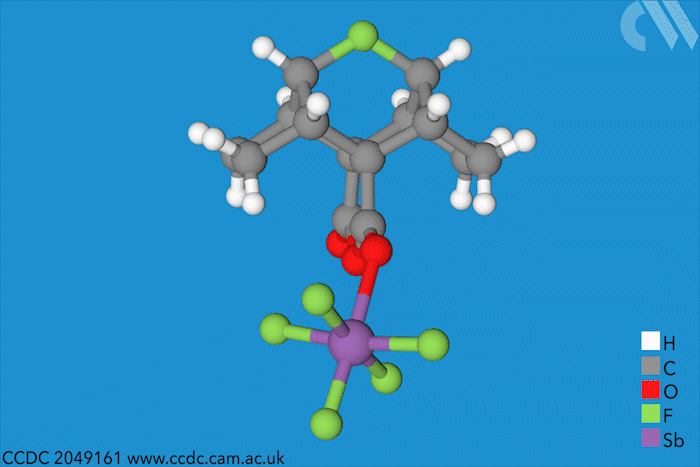
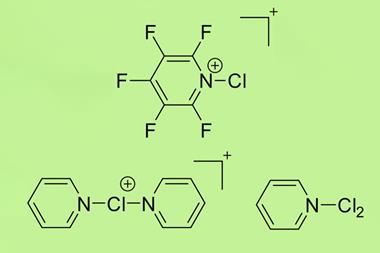
Halonium ions of the type [R2X]+ were discovered as far back as 1937. Since then, many stable iodo-, bromo- and chloronium salts have been made. But fluoronium didn’t make an appearance until 1992, when it showed up as short-lived species in mass spectrometry experiments. Since then, chemists have tried to characterise it to find out whether it is has a symmetric or a rapidly equilibrating carbocation structure – but a solid structure to prove one way or the other remained elusive.
Now, a team of chemists has synthesised the first fluoronium salt that is long-lived enough to grow a single crystal. The double norbornyl structure was made by abstracting a fluoride ion from a precursor with antimony pentafluoride, a strong Lewis acid, at –50°C. When the resulting mixture was kept at –80°C, yellow fluoronium salt crystals formed over the course of two weeks.
X-ray crystallography confirmed the two carbon–fluorine bonds to be nearly symmetrical, although computational analysis showed the bonds to be barely covalent due to the strong repulsion between fluorine’s lone pairs and the carbon–fluorine σ bonds. The researchers suggest that this situation is best described as having charge-shift bonds – electrons aren’t shared or transferred but undergo rapid fluctuations that give rise to bonding interaction.
References
K F Hoffmann et al, Nat. Commun., 2021, 12, 5275 (DOI: 10.1038/s41467-021-25592-6)








No comments yet