Does gold need help from palladium to catalyse coupling?
Spanish scientists have proved that gold alone can catalyse cross-coupling reactions following a claim made last year that palladium impurities in the gold are essential for the catalysis.
Avelino Corma from the Polytechnic University of Valencia and his colleagues used kinetic and theoretical studies to prove their findings, published in 2007, that gold, coupled with cerium oxide, catalyses the reaction alone.1 ’Gold nanoparticles are active enough to promote the reaction, regardless of the presence or absence of palladium,’ says Corma.
A paper by Antonio Echavarren from Spain’s Institute of Chemical Research of Catalonia and colleagues, says that palladium impurities are crucial for the process.2 In their report, Echavarren’s team performed a similar reaction to the one carried out by Corma, coupling phenylacetylene with iodobenzene in a palladium-free setting with a homogeneous gold catalyst complex. They found that gold alone failed to catalyse the reaction that time.
Corma’s findings agree with this, and when his team tested the reaction again, they found that it did only proceed under conditions in which gold complexes decomposed in solution to form gold nanoparticles, after which the Sonogashira coupling occurred.
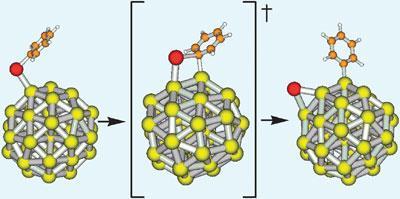
It’s the gold nanoparticles that are crucial, says Graham Hutchings, an expert in heterogeneous catalysis and surface science from Cardiff University, UK. ’Corma’s paper sets the record straight for this interesting reaction. Until the 1980s, everyone would agree that as gold was the most noble of metals, it could not be a catalyst but now we know different.’
’These results are in agreement with work that we carried out on the activations of alkenes and alkynes, in which we concluded that fundamental differences exist between homogeneous and heterogeneous gold catalysts,’ says Echavarren.
’Sonogashira coupling is important because it’s used to prepare compounds for use from electronics to medicine,’ says Corma. But, he adds that the next challenge is to get a better understanding of the reaction mechanism to define the differences between the reaction pathways for gold and palladium. ’It’s necessary to revisit other reactions catalysed by gold to assess the role of nanoparticles and palladium impurities on the overall catalytic activity.’
Elinor Richards
Link to journal article
Gold catalyzes the Sonogashira coupling reaction without the requirement of palladium impuritiesAvelino Corma, Raquel Ju?rez, Mercedes Boronat, F?lix S?nchez, Marta Iglesias and Hermenegildo Garc?a,?Chem. Commun., 2011, 47, 1446DOI:10.1039/c0cc04564k
References
1 C González-Arellano et al, Angew. Chem., Int. Ed., 2007, 46, 1536, DOI: 10.1002/anie.200604746.
2 T Lauterbach et al, Org. Lett., 2010, 12, 3006, DOI: 10.1021/ol101012n.






No comments yet