Hybrid nanoparticle-ionic liquid electrolyte could open the door to safer, more durable batteries with lithium metal anode
Safer, more durable batteries are the aim of a US team that has made a new, hybrid nanoparticle-ionic liquid electrolyte.
Batteries containing lithium metal as the ion source can store the highest known density of energy, but the most commonly used electrolytes (the ion-conducting medium within the battery) do not work well in these batteries as they are unstable in typical operating conditions and may break down disastrously.
This problem has stimulated research into other electrolytes, made of polymers or ionic liquids (ILs). While polymer electrolytes generally have low conductivity at room temperature, ILs are salts in a liquid state and thus have good conductivity and stability under the battery operating conditions. They are, however, unable to prevent the formation of lithium networks called dendrites, which degrade battery performance.
A team led by Lynden Archer at Cornell University, New York has now developed a new family of solventless electrolytes made of hard zirconium oxide nanostructures linked to softer IL species, using a simple one-pot procedure. The electrolytes, which are a gel-like fluid at room temperature, are temperature- and redox-stable which makes them suitable for use in batteries, they transport lithium ions well and they also prevent the growth of dendrites.
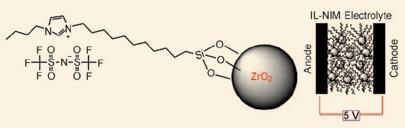
Archer is motivated by making batteries for everyday use, such as for electric vehicles, and says the technology has a nice connection to making very high energy-density rechargeable batteries. ’Because the components are joined by ionic bonds they can’t vaporise easily. From the point of view of safe, explosion-proof secondary batteries, this is in all respects ideal,’ he adds.
Michel Armand, an expert in lithium batteries at University of Picardie in Amiens, France, is less certain. ’It is a very clever material design but it must be tested at currents 100-1000 times higher to show it is suitable for practical operation,’ he says.
Archer believes that tethering the cation part of the IL maximises the lithium ions’ contribution to the conductance of the battery. However Armand points out that the anion part of the IL could be mobile, and extended battery use could result in depletion of the salt from one side of the battery. He suggests that the design could easily be modified to tether the anion also, thereby preventing movement of all but the lithium ions, and potentially making a better battery.
Carol Stanier
References
et alAngew. Chem. Int. Ed., 2010, DOI: 10.1002/anie.201004551






No comments yet