Enzymes buried in the genomes of soil bacteria can be harnessed to modify natural molecules in new ways
US Researchers have harnessed enzymes hidden in the genomes of soil bacteria to modify a natural antibiotic molecule in ways that would be difficult or impossible by traditional synthesis. The technique could be applicable to other families of molecules, providing easy access to a huge variety of complex molecules.
Synthesising novel derivatives of known drugs is a widely used method to find new drug candidates and overcome problems such as antibiotic resistance. The team, led by Sean Brady at the Rockefeller University in New York, chose a family called glycopeptides to test out their idea.
Glycopeptides are defensive antibiotics produced by a range of bacteria and have a cyclic peptide core with sugars and sulfate groups attached at a variety of different positions. ’We chose glycopeptides as model compounds because there are many known derivatives out there,’ says Brady. ’We wanted to show that by can make derivatives that have never been made previously.’
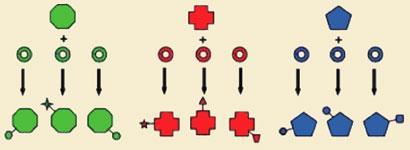
The team extracted DNA in bulk from soil samples and generated large libraries containing up to 15 billion unique DNA fragments. They then searched this environmental DNA (eDNA) megalibrary for sequences corresponding to the proteins involved in making glycopeptides.
Their aim was to find the enzymes that attach sulfate decorations to the glycopeptide core. ’By systematically going through an environmental sample of DNA we were able to find additional glycopeptide-producing biosynthetic gene clusters from organisms in soil. Once identified, we could then use the functionalisation ability of these tailoring enzymes to make a library of modified glycopeptides’, says Brady. The search turned up six unknown tailoring enzymes, which the team used to generate 15 new unnatural glycopeptides with sulfate groups attached at specific positions.
’It’s easier to not to have to build the core structure again and just go and harness from the environment the enzymes that we know are going to be different in each pathway and yet selective towards the core structure’, adds Brady.
In recent years researchers have sequenced complete genomes of many useful natural product-producing organisms. ’We and others were quick to recognise that there were a lot of structurally novel natural products with potentially interesting and useful bioactivity yet to be discovered in such organisms, even very well-studied ones’, says Greg Challis from the University of Warwick, UK, who works on natural product biosynthesis and genome mining approaches to new natural product discovery.
Challis adds that the prevailing view in the pharmaceutical industry 10 years ago was that there was little structural novelty left to be discovered in the reservoir of bioactive natural products. ’Genome sequencing has shown us that this is not true,’ he says. ’Brady’s paper provides a nice example of how the structural variation in a well known class of clinically important drugs can be significantly expanded by tapping into the soil metagenome’.
Future work in Brady’s lab will focus on applying this technique to other families of molecules with a common, functionalised core. ’But we want to look at rarer families’, says Brady.
Akshat Rathi
References
J J Banik et alJ. Am. Chem. Soc., 2010, DOI:10.1021/ja105825a





No comments yet