Chinese chemists optimise process to make useful cyclohexanone from phenol in a single step
A highly efficient route to cyclohexanone - a key chemical in the production of nylon - has been developed by researchers in China. The process uses a dual catalyst of palladium with a Lewis acid, and could greatly simplify production of millions of tonnes of cyclohexanone made each year.
Cyclohexanone is currently made through hydrogenation of phenol - but this process is prone to over-reduction, producing cyclohexanol as a major by-product. A further dehydrogenation step is typically used to bring the mixture back to the desired product.
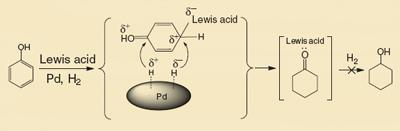
To avoid this additional step, the Chinese team investigated a range of catalysts before settling on a combination of palladium nanoparticles mounted on a solid support of either carbon, zeolite or alumina, and a Lewis acid, such as aluminium chloride. This combination prevents over-reduction, and achieves more than 99.9 per cent conversion of phenol to produce cyclohexanone with 99.9 per cent selectivity.
’The high activity and selectivity of the dual catalyst system can be explained by the synergy of the palladium and Lewis acid for the reaction,’ says Buxing Han, who worked on the project at the Chinese Academy of Sciences in Beijing, China.
’Palladium activates the hydrogen gas, and the Lewis acid makes the benzene ring of phenol more active to hydrogenation,’ explains Han. ’Crucially, once the cyclohexanone has formed, acid-base interactions between the Lewis acid and the ketone group inhibit further hydrogenation to cyclohexanol.’
Thanks to the high selectivity, separating out the final products and recycling the catalyst is a simple process. However, Mark Keane at Heriot-Watt University in Edinburgh, UK, thinks the ten atmospheres of pressure required and seven-hour reaction time may prove tricky to scale-up to the quantities required in industry.
’At the moment, this is a standard organic chemistry approach,’ says Keane. ’For large-scale industry it needs to move away from a fixed-volume process to a continuous flow system. If it can be optimised to work at lower pressures and higher reaction rates, then it has potential to become a remarkable process.’
Another problem Keane notes is the use of dichloromethane as a solvent, but the Chinese team have already taken steps towards solving this problem: ’We are particularly keen to use green chemistry and green solvents,’ Han told Chemistry World. To demonstrate this, they showed that the reaction could be performed more efficiently under high pressure using compressed CO2 as the solvent, and believe that the process can be adapted for continuous production in the future.
Lewis Brindley
References
H Liu et al, Science, 2009, DOI: 10.1126/science.1179713






No comments yet