pH plays a key role in the structure of a naturally-occurring amyloid protein, helping the body control its aggregation
US researchers studying the naturally-occurring amyloid protein Pmel17 have discovered that pH plays an important role in its structure, helping the body control its aggregation.
Misfolded fibrous proteins, called amyloids, are commonly associated with diseases such as Alzheimer’s and Parkinson’s. However not all amyloids are disease related - they can also fulfill useful roles. To determine what marks this difference, Jennifer Lee and colleagues at the National Institutes of Health in Bethseda, US, studied the structure of Pmel17; an amyloid which acts as a scaffold for melanin formation.
’Melanin is formed in an organelle called a melanosome,’ says Lee. ’We were interested how pH solution would affect [Pmel17] amyloid formation because the organelle’s pH changes as it matures.’
Lee analysed the structure of the repeat domain of Pmel17 in vitro. She found that beta-sheet aggregates formed around pH4 and these elongated into fibrous scaffolds at pH5. However at pH values above 6, the amyloid completely dissolved.
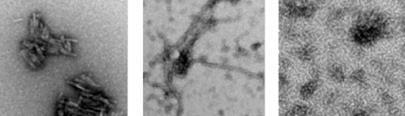
’Pmel17 has a lot of carboxylic acid side chains which are critical in aiding the formation of amyloid structure,’ says Lee. ’It’s very different from disease related amyloids which are quite robust. At physiological pHs Pmel17 fibres would not form so maybe that’s why they’re benign.’
’The striking thing is that the pH range is so narrow,’ says Filip Meersman who researches protein folding and amyloids at the Catholic University of Leuven, Belgium. However he says the work does not appear to have any direct implications for understanding disease-related amyloids.
’What we’re trying to understand is whether it is on the molecular level that the functional and disease related amyloids are different, or is it the way amyloid formation is controlled by the cell?’, says Lee. ’For example if the amyloid formed in these organelles were to leak then they’re not stable under normal cellular conditions. This is the beginning in trying to understand and compare and see if that’s the case.’
Manisha Lalloo
References
C M Pfefferkorn, R P McGlincheyb, and J C Lee, Proc. Natl. Acad. Sci. USA, 2010, DOI: 10.1073/pnas.1006424107






No comments yet