A 21-atom nickel chain is the longest metallic polymer ever created. Stabilised by a macromolecular ligand shell, these one-dimensional molecular wires fuse the durability of plastics with the conductivity of metals and could have significant implications for nanoelectronics.
Metals and polymers are vital materials in our daily lives but the distinct electronic structures of metal and nonmetal atoms mean it is difficult to effectively combine the properties of these versatile materials. Strong non-metal bonds stabilise conventional polymers but act as thermal and electronic insulators, while weak metal−metal interactions preclude the formation of stable metallic chains. ‘The majority of known metallopolymers are based on organometallic repeat units, and do not feature direct metal−metal interactions,’ explains Rebecca Musgrave, a metallopolymers researcher from King’s College London, UK. ‘Although extended metal atom chains have been well studied, these types of compounds are typically limited in length (containing between three and 11 metal atoms), and can best be described as oligomeric species rather than true polymers.’
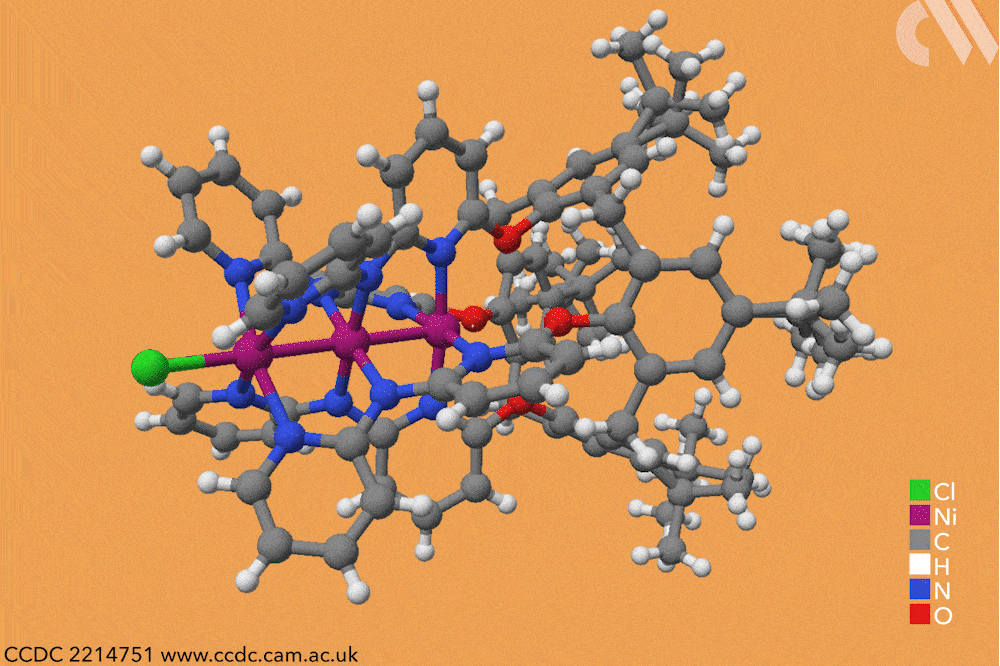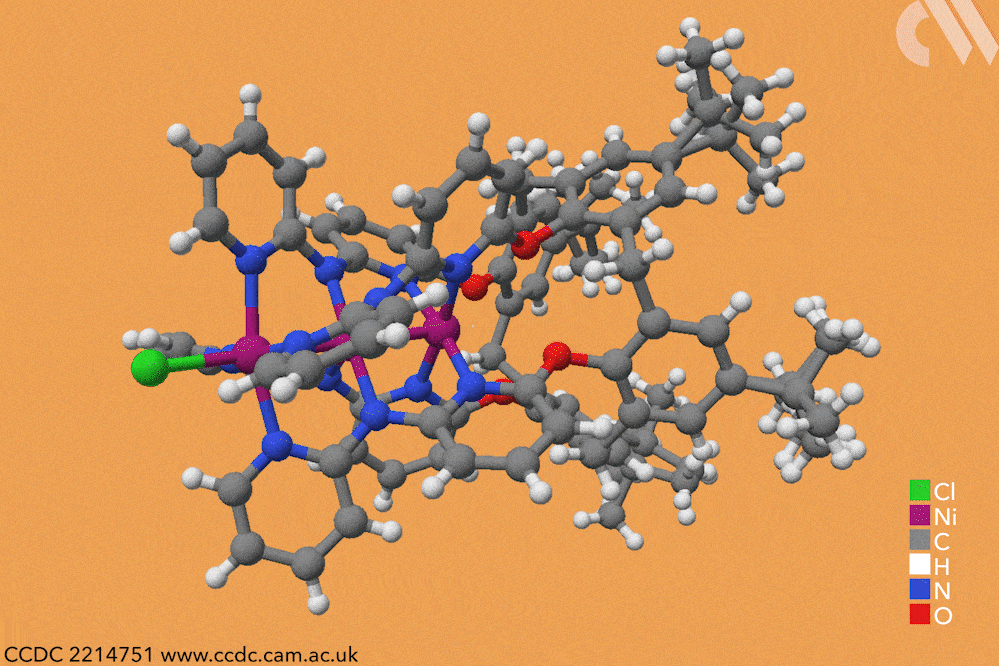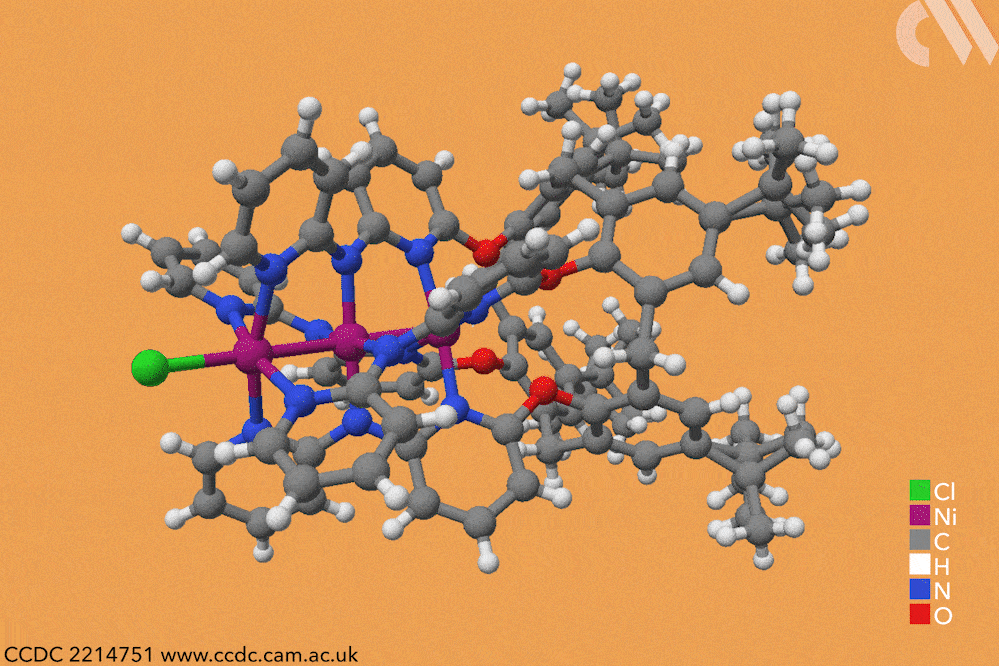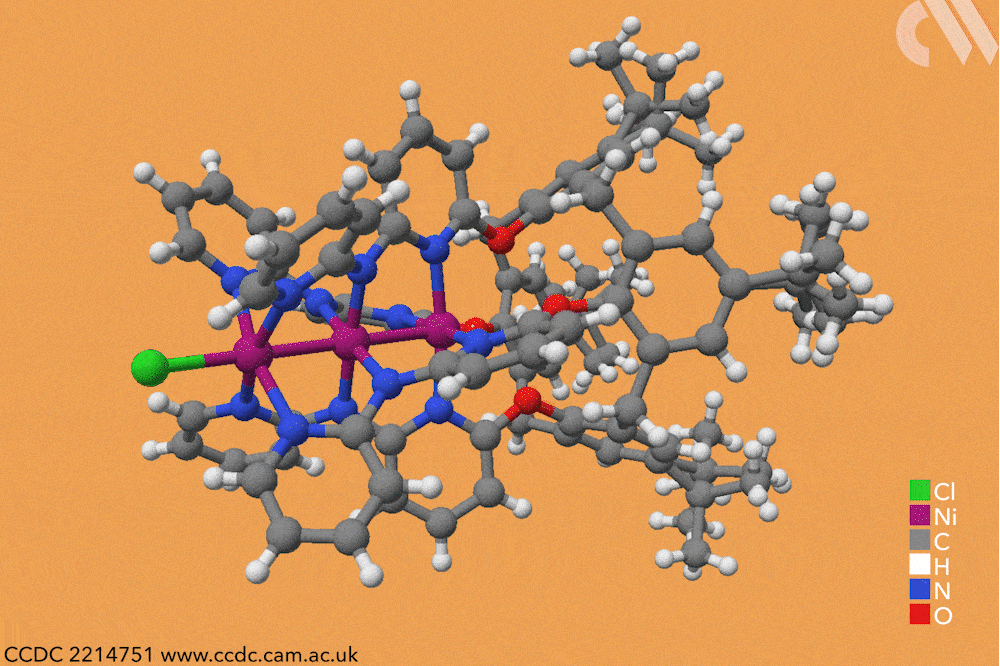
However, using a carefully constructed ligand system, a team led by Guowei Wang and Huisheng Peng at Fudan University in China has successfully constructed a series of metal-backboned polymers up to 21 atoms in length. The team pre-assembled a four-armed macromolecular ligand which acts as a template and protective sheath for the fragile metal chain.
The cup-shaped molecule 4-tert-butylcalix[4]arene acts as a scaffold, and nitrogen-containing ligands are iteratively added along each of its four arms to build up the stable skeleton of the polymer. Upon subsequent exposure to nickel acetate, metal atoms coordinate to these nitrogen-containing ligands, forming a chain of nickel atoms within the tube-like cavity of the macromolecule. By controlling the number of nitrogen ligands in this macromolecule, the team were able to vary the length of the metallic chains from three to 21 atoms and produce a series of polymers with graded properties.
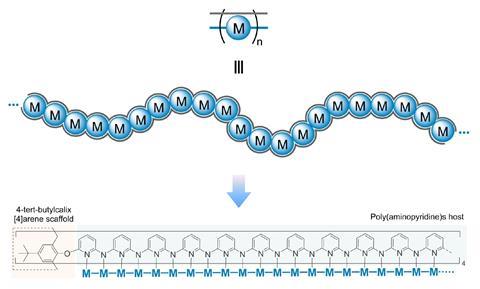
Keen to explore the characteristics of these new materials, Wang and Peng conducted a series of spectroscopic and computational studies. X-ray analysis confirmed the unusual structure – a linear chain of nickel atoms, each coordinated to four nitrogen ligands and two metallic neighbours – but also revealed an interesting structural trend. The nickel−nickel bond length, already noticeably shorter than in the bulk metal, decreased as the length of the nickel chain extended, resulting in a stronger structure in longer chains. The authors believe that the unusual bonding of the metal atoms allows electrons to delocalise over the entire metal framework, an essential property for conductive materials. Correspondingly, the team also observed greater electronic conductivity in longer chain polymers.
‘The tunability of electronic properties makes these materials promising candidates in semiconducting applications, as well as in the area of “molecular wires”,’ comments Musgrave. ‘It would be fascinating to understand the electron transport properties of these polymers by performing molecular conductance experiments.’
Wang and Peng are continuing to design a variety of metal-backboned polymers to explore the potential of these materials and are confident that this synthetic strategy will both extend to other transition metals, and enable other researchers to develop new functional polymers.
‘The elegant pre-assembly of the macromolecular ligand structure has allowed Wang and Peng to template one-dimensional metallic chains of impressive proportions with excellent control,’ says Musgrave. ‘It’s beautiful work!’
References
K Zeng et al, Angew. Chem., Int. Ed., 2023, DOI: 10.1002/anie.202216060















No comments yet