Quantum dots reveal dynamics of viral infection.
Glowing quantum dots are helping researchers study how viruses infect cells, and although the fluorescent nanoparticles have only been used on plant viruses so far, the technique could prove to be invaluable for drug development.
The dots are made of tiny crystals of cadmium selenide, surrounded by a shell of zinc sulfide. Bogdan Dragnea, a chemist at Indiana University, Bloomington, US, leads the team that encapsulated these dots inside the protein coat that surrounds a virus, called the viral capsid.1
The great thing about quantum dots, said Dragnea, is their ’remarkable photostability and brightness’. They shine brightly for a sufficiently long time (up to 10 minutes) to track what a virus is up to. The passage of the encapsulated quantum dots into a cell can be tracked using a confocal microscope, which produces sharper images than conventional microscopes by using a pinhole to exclude out-of-focus light.
Dragnea’s team adapted a technique they had developed to get gold nanoparticles inside viral capsids.2 The nanoparticle-capsid complex was built up by a self-assembly process, driven by the attraction between negatively charged gold nanoparticles and positively charged proteins that line the viral capsid. They believe this mimics the interaction between the anionic genetic contents and cationic protein coat in a real virus.
The researchers reasoned that other components, such as the glowing quantum dots, could be encapsulated like this if they had the same surface chemistry as the gold nanoparticles. Easier said than done.
Get your coat
Dragnea’s team studied four possible quantum dot coatings to find one that was suited to viral encapsulation. Using the wrong coating, or no coating at all, could make the nanoparticles insoluble, or alter the pH of the virus contents, which would halt the assembly process.
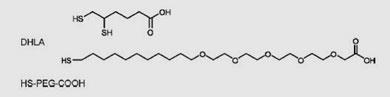
The researchers settled on a coating based on the long, floppy molecule polyethylene glycol (PEG), modified to carry a sulphur atom at one end and a carboxylic acid group at the other. This coating prepares the quantum dot for viral encapsulation, and also keeps the dot fluorescing for a relatively long time. The brightness of quantum dots coated with dihydrolipoic acid, for example, faded about eight times faster than the brightness of the PEG-coated dots.
The viral capsids they studied were based on a well-understood plant virus, brome mosaic virus (BMV). It’s a useful starting point, said David Wright, associate professor of chemistry at Vanderbilt University, Nashville, Tennessee, US, although the technique will be more useful when it can be applied to less well understood, clinically relevant viruses.
But it could be invaluable for studying how long it takes for a particular virus to get across a cell membrane, or what route the virus takes, said Wright, who has stuck quantum dots to the outer coating of viruses to detect their presence in clinical samples.
Dragnea’s technique now needs to be applied to a range of viruses, said Wright: ’it’s really going to be important to make it generalisable.’ Dragnea is now collaborating with biologists at Indiana University to study alphaviruses, which are responsible for a range of mosquito-borne human diseases.
Bea Perks
References
10.1021/nl061165u)
B Dragnea et al, 2003, J Am Chem Soc, 125, 6374






No comments yet