Zoe Schnepp from the University of Birmingham in the UK and colleagues at the National Institute for Materials Science in Japan wanted to show that you don’t have to compromise on properties by being green.
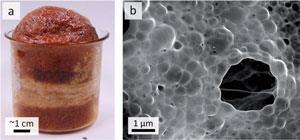
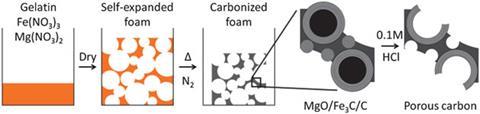
To make the catalyst they first mix iron and magnesium compounds into gelatin to make the gelatin foam. The combination and ratio of these compounds was chosen to create very small bubbles and give a material with a very big surface area. The jelly foam is then heated to 800°C in a furnace. A bubble structure remains but it is now covered in iron carbide. Hydrochloric acid is then used to remove the iron carbide particles increasing the surface area even more by adding an extra dimension of nanopores. ‘You have the big pores from the initial foam of the jelly and you have the nanopores from dissolving out the iron. Together they make a very large surface area that is a very active catalyst,’ says Schnepp. Once it’s dry it looks more like honeycomb than a blancmange.
‘This simple synthesis route may provide an easy and effective recipe for wide use of porous multi-component nanocomposites,’ comments An-Hui Lu, an expert in nanostructured catalyst materials at Dalian University of Technology in China. ‘The resultant material exhibited remarkable performance in oxygen reduction reactions. Although the onset potentials of the doped carbons are slightly higher than a commercial carbon supported platinum catalyst, its good electrocatalytic efficiency and stability, and relatively low cost make it a promising candidate for the next generation of cost-effective oxygen reduction reaction electrocatalysts, particularly for proton exchange membrane fuel cells,’ he adds.
‘It is exciting and highly important research as it not only addresses the scientific community but is also of interest to the public because it deals with a major challenge of modern life – securing an affordable energy supply while sparing the environment at the same time,’ says Nina Fechler, a porous carbon expert from the Max Planck Institute of Colloids and Interfaces in Germany.
Nanoporous carbon expert José Figueiredo, at the University of Porto in Portugal, is a little more cautious and says further work is needed to identify parameters controlling the pore sizes and their connectivity. ‘This work might represent an important step towards the goal of reducing the costs of the catalysts currently used in fuel cells, which has been identified as one of the main reasons delaying the introduction of these devices into the market,’ he concludes.






No comments yet