Ultra-slim silicon membranes: from labs on chips to artificial kidneys.
Tough, ultra-slim silicon membranes could drastically improve the performance of lab-on-a-chip micro-analytical systems, kidney dialysis machines and, in the future, even produce an artificial kidney, claim researchers.
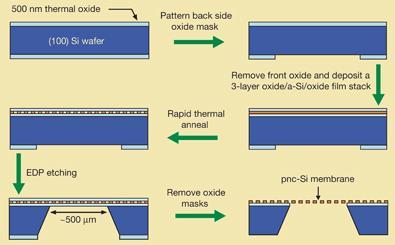
Christopher Striemer, Philippe Fauchet and colleagues at Rochester University New York, US, fashioned the new 15 nm-thick porous nanocrystalline silicon (pnc-Si) membranes using standard semiconductor micro-fabrication techniques.
Compared with commercial ultrafiltration and dialysis membranes, the new silicon films speed up protein separation by at least an order of magnitude.
The secret is fine tuning the size of miniscule pores produced as silicon crystallises during the manufacturing process. ’We discovered it by chance while looking at how thin films of amorphous silicon crystallise when sandwiched between silicon dioxide layers.’ Striemer told Chemistry World. ’You need to control the silicon crystallisation temperature.’

The pores, averaging from 9 to 30 nm in diameter, are formed during rapid thermal annealing from spontaneously-produced voids as silicon nanocrystals nucleate and grow in the 15 nm thick amorphous films. The voids span the whole thickness of the membrane, producing the pores. The pnc-Si thin films are surprisingly tough, able to withstand around an atmosphere of pressure without plastic deformation or cracking.
The new membranes rapidly and efficiently separate proteins from small molecules, different sized proteins under physiological conditions, and can separate similar sized molecules carrying different electrical charges. To demonstrate, the Rochester team chose two common blood proteins with slightly different molecular weights and sizes - bovine serum albumin (BSA) and immunoglobulin-gamma (IgG) - each fluorescently labelled with different dyes. Using real-time fluorescence microscopy, the researchers monitored their passage through two separate membranes with different average pore diameters.
Compared to the hours-long separation times required by commercial dialysis membranes, the smaller pore-diameter silicon membrane completely separated BSA protein from much smaller free dye molecules in little over 6.5 minutes. In the same time, the larger-pore membrane achieved a greater than fourfold separation of the BSA and IgG proteins. Once they thoroughly optimise membrane pore diameters, however, the Rochester team expect to produce even better membranes that selectively exclude IgG, but allow the passage of BSA. Also, by modifying the electrical charge on the membranes’ surfaces, the team selectively blocked dye molecules carrying similar charges.
The new membranes have many potential applications. ’Standard separation techniques are not easy to miniaturise, making protein purification, for example, difficult for today’s lab-on-a-chip systems,’ said Striemer. ’Our pnc-Si membranes could be directly inserted into such micro-fluidic systems, making separation steps potentially simple and highly efficient.’
But the new membranes could have far-reaching bio-medical applications. Given their efficiency, Striemer predicts pnc-Si membranes could greatly simplify dialysis systems, and even lead to better patient outcomes.
And an artificial kidney? ’Practically impossible, using commercial polymer dialysis membranes’ said Striemer. ’But our material might prove useful here. Our primary concern with artificial organs is the issue of long-term bio-stability for our 15 nm thick material. We just don’t know, and regulatory hurdles in the US would make this very difficult. But in principle, why not?’
Lionel Milgrom
References
C Striemer, et al, Nature, 2007; 445, 749






No comments yet