Unusual spin state could open the door to new and exciting chemistry
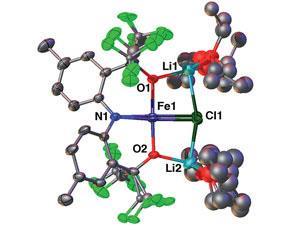
University chemistry students are taught that the shapes and electronics of inorganic complexes are predictable. For example, d8 square-planar Pd(ii) and Pt(ii) complexes are invariably low spin, while d3–d7 tetrahedral complexes are high spin. Now, researchers in the US have thrown away the textbook by synthesising a square-planar Fe(ii) complex that is not only high spin, but has a different core (FeO2NCl) to the only other examples of this complex type, all of which feature an FeO4 core.
Adam Veige at the University of Florida, Gainesville, and colleagues made use of an easily modifiable pincer ligand they had previously developed to make the unusual complex. They also provided conclusive evidence via high field EPR and Mössbauer spectroscopy that these unique features are maintained in both the solid state and solution.
Convincing metal ions to adopt unusual geometries and electronic structures like this is often the first step to discovering new and exciting chemistry. The team’s findings could have particular significance as iron complexes have become increasingly important as less toxic, cheaper alternatives to complexes based on noble metals for many applications.
References
This article is open access. Download it here:
M E Pascualini et al, Chem. Sci., 2014, DOI: 10.1039/c4sc02634a
![An image showing a single-crystal X-ray derived model of [MH6] complex](https://d2cbg94ubxgsnp.cloudfront.net/Pictures/380x253/4/0/6/501406_full_text__crimmin_161621_859886.jpg)
![An image showing the molecular structure of the [FeFe]-hydrogenase active site, the H-cluster](https://d2cbg94ubxgsnp.cloudfront.net/Pictures/380x253/1/9/3/501193_index_834443.jpg)






No comments yet