A one step self-desymmetrising reaction forms complex molecules from simple substrates.
Researchers have found a way to synthesise complex molecules in one step using simple and easily synthesised substrates.
In a remarkable process, Robert Stockman and colleagues from the University of East Anglia, UK, and the University of Amsterdam, the Netherlands, combined two-directional synthesis with a tandem reaction to produce a non-symmetrical quinolizidine from a symmetrical, acyclic mesylate.
Two-directional synthesis, which generates symmetrical products from symmetrical starting compounds, led to a product with two identical chains. These were then folded in just a few steps by a tandem reaction to form a polycyclic compound.
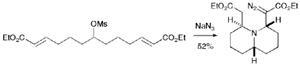
The strategy investigated by the team is designed to reduce the number of steps required in the synthesis of complex molecules. The process involved tandem azide formation, intramolecular cycloaddition, triazoline fragmentation and Michael addition.
’With diversity oriented synthesis requiring a host of unique and complicated molecular frameworks, I envisage the concept of combining two-directional synthesis and tandem reactions may provide a useful access point for those wishing to generate such molecular scaffolds,’ said Stockman.
The future holds further challenges for Stockman and his co-workers as they continue to develop novel and unusual tandem reactions for use in diversity oriented synthesis and target based synthesis.
Elinor Richards
References
M Rejzek, R A Stockman, J H van Maarseveen and D L Hughes, Chem. Commun., 2005, 4661 (DOI: 10.1039/<MAN>b508969g</MAN>)






No comments yet