Using an aqueous cathode increases capacity and avoids wear during charging cycles, researchers say
A new approach to alkali batteries, in which the cathode is dissolved in water that flows through the system, could overcome the limitations of currently available batteries, scientists in the US say. Rechargeable lithium-ion batteries have revolutionised the mobility of small electronics, but their limited capacity means they don’t stretch easily to bigger jobs such as running automobiles or storing power for national grid networks.
Ordinarily, lithium ion batteries contain a solid anode, which gives up electrons, and a solid cathode, which receives them. The two are divided by an organic fluid electrolyte. This strategy depends on a cathode that can expand to host additional lithium ions from the anode during discharge and contract to release them during charging. The solid’s limited ability to change its volume caps the battery’s capacity, and the expansion and contraction is also a source of wear.
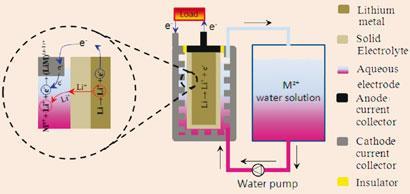
Aqueous cathodes in ’flow through’ batteries are not a new idea, but Yuhao Lu, John Goodenough, and Youngsik Kim at the University of Texas in Austin, US, are the first to suggest them as the next phase of lithium-ion technology. Their design places a solid alkali ion electrolyte between a lithium or sodium anode and an aqueous cathode comprising a metal - in this case iron - in two oxidation states. The cathode solution flows through the system from an attached storage tank, so its total capacity can be much greater than that of a solid. Moreover, because the cathode is a liquid, it avoids the volume changes as lithium shuttles in and out.
To test the idea, the team used a commercial electrolyte called lithium superionic conductor (Lisicon) with a solid lithium anode. An iron cyanide cathode met many of the requirements for a successful battery: it’s chemically stable, it’s soluble in water and its high potential of 3.4V with lithium means it is on par with commercially available lithium-ion alternatives.
The electrolyte restricted their initial experiments to a proof of concept only, says Goodenough. But a thinner, more stable lithium ion solid electrolyte is already under development in his laboratory.
Rob Armstrong of the University of St Andrews in the UK describes the work as ’highly significant’. Although the solid electrolyte requires improvement, he says, ’the suitability for both static energy storage associated with renewable energy generation, and for electric vehicles, means that this represents a very promising avenue for future research, alongside lithium-air and lithium-sulfur batteries’.
Kate McAlpine
References
Y Lu, J B Goodenough and Y Kim, J. Am. Chem. Soc., 2011, DOI: 10.1021/ja201118f






No comments yet