Meera Senthilingam
This week, we go back to the wartime to discover an agriculturally crucial compound, with Lars Öhrström…
Lars Öhrström
On the 28th of July 1942 the UK War Cabinet met in, or probably under, Whitehall, and the brief minutes you can now download from The National Archives includes an alarming statement by the minister of agriculture Robert Hudson: ‘We haven’t enough in [the] country to last beyond Christmas.’
What did they not have enough of? Phosphates. These are a wide range of chemicals you make by subtracting one or more protons from this week’s compound, phosphoric acid, H3PO4, and they are especially important for producing fertilisers. The minister was worried because he needed to keep the agricultural production up in a country that prior to the war had imported a large part of its foodstuffs and was then blockaded by enemy shipping. Phosphate fertilisers were critical for this effort on the agricultural ‘home front’.

In the minutes, Hudson states one of the causes of his headache: ‘apatite from North Russia is off!’ Apatite is the raw material for phosphates, a calcium hydroxy phosphate mineral known as phosphate of lime, with formula Ca5(OH)(PO4)3, and it needed to be imported. One possibility was to ship it in from Russia on the homecoming leg of the famous Arctic Convoys to Archangelsk and Murmansk. These had, however, been temporarily suspended after the infamous PQ17 convoy lost 24 of its 35 ships earlier the same month, and it was unclear when they could be resumed.
Phosphoric acid easily qualifies to the top-ten list of the industrially produced chemicals year after year and it’s mainly used to make fertilisers, now just as it was in the 1940s. It has a central phosphorus atom surrounded by four oxygens atoms in a tetrahedron, and this unit, known as the phosphate ion, has a charge of minus three. To complete the molecule there are protons, or H+ ions, attached to three of the oxygens atoms.
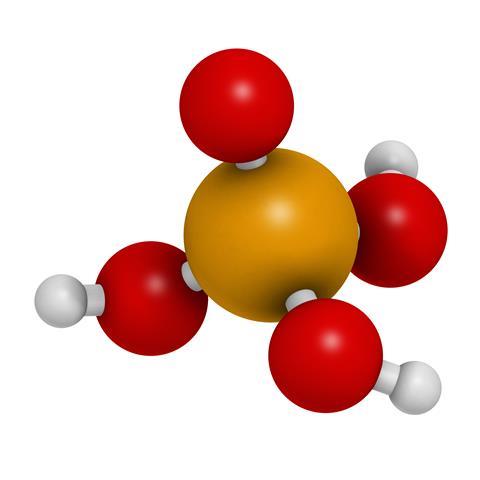
Through its cycle in the environment and the body, this central phosphate part does not change much. When the acid is dissolved in water at neutral pH we mainly have the dihydrogen phosphate anion H2PO4– and the monohydrogen phosphate dianion HPO42– swimming around in the solution. But when it is incorporated into various biological functions, it is normally in the form of the phosphate ion itself, with no hydrogen atoms and three negative charges. What are, then, these vital biological functions that make this molecule so important for us and for agriculture?
Together with calcium ions it reverts back to apatite in our bodies, giving us a skeleton and teeth. We may think of our bones as calcium based, but the truth is that, of the inorganic part, calcium is only 40% by weight and the phosphate ions make up almost 60%.
But wheat and the other grains that concerned the minister of agriculture do not have much of a skeleton, and even carnivorous plants lack teeth, so there must be something more to the biology of phosphate than this. And there is: this anonymous little ion plays a vital part in both our energy system and our information system.

Nowadays we all know about DNA, but even if you know what this abbreviation stands for, deoxyribonucleic acid, you may not be able to decode the role of the phosphate ions as it is quite well hidden. The ‘acid’ in DNA is actually phosphoric acid, but it has replaced two protons with carbon atoms from two different sugars – the ‘ribose’ part – and at physiological pH the third proton is lost as well. It is the back-bone of DNA, holding the long chain together with covalent bonds, but it does not transmit any genetic information. This is the work of the organic nitrogen-containing bases that point inwards into the double helix.
If you take a monomer of the DNA chain, containing ribose, phosphate and just one of the four organic bases, adenine, and add two more phosphate units to the first one, you get a molecule called adenosine triphosphate, or ATP. This molecule is the universal energy currency of the biological world. When it reacts with water and loses one phosphate ion, a process called hydrolysis, energy is released. By intricate mechanisms in our enzymes this energy can be used to power innumerable processes in all living creatures. Most of the fat, sugars and carbohydrates we consume every day, when not stored away in one form or the other, are used to build up our ATP molecules.
It is fair to assume that almost all of the fat, sugars and carbohydrates digested in Great Britain during the war were metabolised to give ATP, because few people grew fat during those years, even though apatite imports from Russia eventually became possible.
Meera Senthilingam
Lars Öhrström, from the Chalmers Tekniska Högskola, in Sweden, with the fertilising chemistry of phosphoric acid. Now, next week, we toast to good health, en français…
Phillip Broadwith
French people have a remarkably low rate of coronary heart disease, despite eating large amounts of saturated fat in the form of butter, cheese, cream and eggs.
This effect is attributed variously to all sorts of factors, but one that crops up time and again is its correlation with drinking red wine. But what is it about red wine that gives it these mystical health properties? Is it the alcohol? Perhaps it could be the antioxidants or tannins from the red grape skins? Is it even the wine at all – is it simply correlation rather than a causal link?
Meera Senthilingam
To find out, join Phillip Broadwith in next week’s Chemistry in its Element. Until then, thank you for listening, I’m Meera Senthilingam.













No comments yet