Conventional wisdom suggests that nanosized bubbles should barely exist at all, so their stability for hours or days has surprised many. Philip Ball takes a close look at these minute miracles

You might call it a fuss over nothing. Nanobubbles are smaller than a bacterium, and empty except for the tiniest puff of gas. But what a furore they’ve caused.
The reason for the controversy is simple: they should not exist. These tiny blebs pocking the surface of a solid immersed in water, too small to be seen in an ordinary optical microscope, seem to defy the usual laws of liquids. They should vanish in an instant, yet they persist defiantly for weeks. Even now, over a decade since they were first sighted, no one really knows why1.
Some researchers think they are a mirage, others an artifact of the experiments mounted to study them. Or perhaps they are an exotic form of matter, not quite vapour but not liquid either. Or maybe they are armour-coated with molecules that protect against their own demise. Some suspect that nanobubbles could be ubiquitous, decorating not just aqueous surfaces but bulk liquids and perhaps constantly popping up in our cells – where they might have biological consequences. In any event they might turn out to have useful properties, killing tumour cells or storing gases – so nanobubbles are a puzzle well worth solving.
Breaking the law
Nanobubbles were first proposed in 1994 by Phil Attard at the Australian National University in Canberra and his colleagues.2 They were trying to understand the deeply puzzling fact that two hydrophobic surfaces in water attract one another over rather long distances: several hundreds of nanometres, which is much greater than the usual reach of intermolecular forces. Attard and colleagues suggested that bubbles a few tens or hundreds of nanometres across – too small to see – might be attached to the surfaces, perhaps formed from dissolved gases. As the surfaces approach, such a bubble might suddenly bridge them, and the surface tension of the bridging menisci would then pull the surfaces together.
The idea finally got what looked like experimental verification in 2000, when two groups – Ko Higashitani and colleages at Kyoto University in Japan,3 and Shi-Tao Lou of the Chinese Academy of Sciences in Shanghai and coworkers4 – independently reported isolated, circular features with hemispherical cross-sections visible with an atomic force microscope (AFM) on solid surfaces under water, which they interpreted as individual nanobubbles.
The only problem was that this did not make sense. To be stable, a bubble has to be in mechanical equilibrium: the pressure on the inside must balance that on the outside. This does not mean that the two pressures themselves are equal: rather, the internal pressure is greater than the external by an amount proportional to the liquid surface tension and inversely proportional to the bubble radius, a relationship first deduced by Thomas Young and Pierre-Simon Laplace in the early nineteenth century. So the smaller the radius, the greater the internal pressure. For a nanobubble this implies that the internal pressure should be huge – which in turn means that the gas inside should rapidly diffuse across the interface into the water, and the bubble will dissolve. A 100-nm-radius bubble would, on this basis, be expected to survive for less than a millisecond. Yet here they seemed to sit, visible for hours, days, even longer.
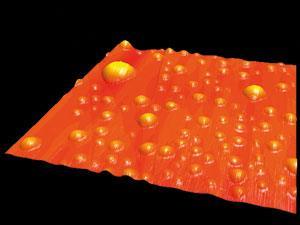
For this reason, the early observations of nanobubbles were widely dismissed. It was suspected that the blobs visible in the AFM must be some kind of surface contaminant. Among the sceptics was chemical engineer William Ducker, an expert on surface forces then working at the University of Melbourne in Australia, and now at Virginia Tech in Blacksburg. In 2007 he set out to, in his own words, ‘prove that they didn’t exist’. With his colleague Xuehua Zhang he used spectroscopy to probe the state of the stuff these putative nanobubbles were made of.5 If it was really a gas, they should be able to see the spectroscopic fingerprint of the molecules rotating. But if the features were some surface contaminant, any rotation should cease or at least be very hindered.
To their surprise, Ducker and Zhang found that the nanobubble material indeed appeared to be gaseous, and surprisingly had a pressure close to atmospheric, rather than being much higher. They had no choice but to conclude that nanobubbles do exist after all. ‘In the end one has to change one’s opinion when the evidence points so obviously in the other direction’, says Ducker. According to Vincent Craig at the Australian National University, these measurements were ‘the final piece of evidence that people needed’. Zhang says that ‘after we published this paper, more and more researchers began to accept that surface nanobubbles are real’.
Survival strategy
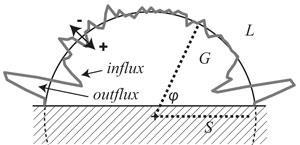
So how do they survive? Ducker has suggested that the bubble’s air–water interface should act as a kind of magnet for all sorts of dissolved contaminants: any hydrophobic molecules ought to get lodged there, just as hydrocarbon molecules accumulate as a surface film in water. This coating would both lower the surface tension, which according to the Young–Laplace equation also reduces the pressure difference inside and outside the bubble, and also reduce the rate of gas diffusion across the interface.
But Detlef Lohse, an expert in fluid dynamics at the University of Twente in the Netherlands, thinks that these surface nanobubbles are not in equilibrium at all. Rather, he suggests that they are constantly being fed with dissolved gas to replenish that lost by dissolution, like a lake kept in a steady state as incoming streams replace water lost by evaporation. In collaboration with Michael Brenner at Harvard University, Lohse has developed a theory in which gas is adsorbed in a thin film on the surface of a submerged hydrophobic surface and flows laterally into the nanobubble, while gas molecules in the bubble constantly escape into the liquid.6 ‘The idea is that the outflux of gas through Laplace pressure is compensated by some influx close to the contact line of the bubble and the surface,’ Lohse explains. This model suggests that nanobubbles should have a preferred size: ones smaller than this size will grow, and larger ones will shrink. Such a preference is indeed seen in some experiments, but not all.
Both Craig and Zhang admit to being uncomfortable with all of the current explanations for nanobubble stability. ‘I can’t see a thin layer of contaminant being able to completely prevent gas transfer,’ says Craig. Besides, Ducker himself, in collaboration with Zhang and others, has recently reported that adding a surfactant to a solution supporting nanobubbles on graphite does not alter their stability. Yet the surfactant would be able to ‘clean’ the solution by mopping up any organic contaminants. ‘This really casts some doubt on my hypothesis,’ Ducker admits.
Lohse is more definitive: ‘I think that there is a consensus that it is not contamination that is the answer,’ he says. However, his dynamical explanation has problems too. In particular, it’s hard to understand how the flow of gas in and out of the bubbles could be sustained for very long periods. ‘For the model to work, the bubble volume has to turn over at a rate of about 1 million times per second,’ says Craig. ‘My intuition isn’t comfortable with this.’ Neither is he persuaded by a third explanation, recently advanced by Zhang and others, which argues that the edge of the nanobubble, where it meets the surface, gets ‘pinned’, so that any gas loss would reduce the curvature and stabilize rather than destabilize the bubble. ‘I wish I could offer another explanation,’ Craig admits ruefully. Ducker wonders whether the Young–Laplace equation itself breaks down a these very small scales. At the moment, however, it remains a mystery why nanobubbles persist.
Bubbles in space
So far, most work on nanobubbles has studied them at solid surfaces. But more recently researchers have started to wonder whether, if they can be somehow stabilized here against the destruction that the Young–Laplace equation seems to threaten, might that not happen in bulk solution too? Can free-floating nanobubbles form and survive?
In 2010 Kazunari Ohtaki at Osaka University in Japan and his colleagues reported the first apparent evidence that they can. They supersaturated water with nitrogen, methane or argon, agitated it and then froze it very rapidly. Electron microscopy of the fractured icy samples showed that they were full of bubble-like voids about 100 nm across, even after the solutions had been left to stand for two weeks.8 Although this work remains controversial, others have since claimed to see nanobubble-like features in freeze-fractured aerated water, including the wastewater outflow from a sewage plant.
Meanwhile, Craig has come across tantalizing evidence in the old literature that bulk nanobubbles might be being produced all the time in liquids. In 1962 two nuclear scientists in Italy, Daniele Sette and Franco Wanderlingh, were looking at the use of ultrasound to make bubbles – ordinary, visible bubbles – in water. They found that if they placed a lead screen around their sample, the threshold energy needed to induce cavitation and make bubbles rose significantly – but only gradually over a period of ten hours or so.9 The lead screen was preventing cosmic rays, specifically cosmic-ray neutrons, from entering the sample. But if the neutrons were themselves triggering the formation of bubbles, the effect should be more or less instantaneous. Perhaps, Craig suggests, the neutrons were instead nucleating nanobubbles with long lifetimes, which in turn seeded bubble formation by ultrasound.
If it is hard to figure out how surface nanobubbles can be stable, bulk nanobubbles pose even more of a challenge. For one thing, bulk nanoubbles will be spherical rather than hemispherical caps, so they’ll be even more highly curved than surface nanobubbles for the same volume. And there is typically not enough organic contaminants to ‘armour’ the high concentrations of nanobubbles seen by Ohgaki and colleagues. But Lohse and his colleagues James Seddon and Joost Weijs have proposed that high concentrations of bulk nanobubbles can ‘shield’ one another by keeping the surrounding liquid highly supersaturated with gas, slowing down the mutual rate of gas loss.10 ‘Beyond a certain concentration, it is energetically cheaper for the gas molecules to cluster together than to dissolve,’ Lohse explains. Their computer simulations support this idea.
Bubbles in action
If cosmic rays can nucleate nanoubbles, we ought to have them in our oxygenated bloodstream. But wouldn’t we have noticed? ‘If we’re walking around with nanobubbles inside us, I don’t think it would really cause much of a problem because they seem to be so inactive and immobile,’ say Craig. Yet there’s one possible exception to that: decompression sickness, where nanobubbles could nucleate larger, more problematic, ones. ‘Decompression sickness symptoms appear more strongly in the part of the body that has been exercised,’ says Craig. ‘So it’s possible that small muscle tears lead to the production of hydrophobic surfaces that can sustain nanobubbles and later lead to decompression sickness.’
This and other possible biological roles of nanobubbles1 remain speculation at present. However, if they can be produced to order on surfaces or in solution, they could find many uses. Nanobubbles stabilized with polymers have been used as little capsules to transport the anticancer drug doxorubicin into tumours, where the drug was released by bursting the bubbles using ultrasound. And the energy released by bursting nanoubbles generated using lasers was used in 2010 by a team at Rice University in Houston, Texas, to damage tumours in fish.

If they are real, bulk nanobubbles would offer a great way to load up water with gas. Gang Pan of the Chinese Academy of Sciences in Beijing hopes to exploit that to deliver oxygen to natural waters, where it has been depleted by algal blooms caused by eutrophication. Pan has developed a method of loading particles of porous natural clay with oxygen nanobubbles. When dumped into the lake, the clay then sinks to the bottom and slowly releases oxygen. The method is much less energy-intensive, and more effective, than pumping oxygen directly down to the lake bed, says Pan. He has demonstrated its potential to reverse anoxia in Lake Taihu, near Shanghai, regenerating the clear, blue beauty of the water.11 ‘The Chinese local government has already invested in developing this technique for water-resource protection,’ Pan says.
So perhaps we do not need to fully understand the seemingly miraculous stability of nanobubbles before we can make use of them. All the same, the better we do know how and why they form, the more we are likely to be able to control and tailor that process for useful ends. It is well worth getting under the skin of these mysterious little beasts.
References
1. J. R. T. Seddon, D. Lohse, W. A. Ducker & V. S. Craig, ChemPhysChem 2012, 13, 2179 (DOI: 10.1002/cphc.201100900)
2. J. L. Parker, P. M. Claesson & P. Attard, J. Phys. Chem. 1994, 98, 8468 (DOI: 10.1021/j100085a029)
3. S.-T. Lou, Z.-Q. Ouyang, Y. Zhang, X.-J. Li, J. Hu, M.-Q. Li, F.-J. Yang, J. Vac. Sci. Technol. B 2000, 18, 2573 (DOI: 10.1116/1.1289925)
4. N. Ishida, T. Inoue, M. Miyahara & K. Higashitani, Langmuir 2000, 16, 6377 (DOI: 10.1021/la000219r)
5. X. H. Zhang, A. Khan & W. A. Ducker, Phys. Rev. Lett. 2007, 98, 136101 (DOI: 10.1103/PhysRevLett.98.136101)
6. M. P. Brenner & D. Lohse, Phys. Rev. Lett. 2008, 101, 214505 (DOI: 10.1103/PhysRevLett.101.214505)
7. X. H. Zhang, X. Zhang, J. Sun, Z. Zhang, G. Li, H. Fang, X. Xiao, X. Zeng & J. Hu, Langmuir 2007, 23, 1778 (DOI: 10.1021/la062278w)
8. K. Ohgaki, N. Q. Khanh, Y. Joden, A. Tsuji & T. Nakagawa, Chem. Eng. Sci. 2010 65, 1296 (DOI: 10.1016/j.ces.2009.10.003)
9. D. Sette & F. Wanderlingh, Phys. Rev. 1962, 125, 409 (DOI: 10.1103/PhysRev.125.409)
10. J. H. Weijs, J. R. T. Seddon & D. Lohse, ChemPhysChem 2012, 13, 2197 (DOI: 10.1002/cphc.201100807)
11. G. Pan et al., J. Lake Sci. in press












No comments yet