Rebecca Trager examines an emerging industry that is growing ‘meat’ outside of animals using cell lines cultivated in bioreactors
More than 90 years ago, Winston Churchill envisioned growing meat in a lab to create a more sustainable food industry. ‘We shall escape the absurdity of growing a whole chicken in order to eat the breast or wing, by growing these parts separately under a suitable medium,’ he wrote in his 1931 essay Fifty Years Hence, imagining synthetic food as ‘the future’ and projecting ahead to the 1980s. Churchill was spot on, though it has taken decades longer than he expected for the science to catch up with his vision.
About nine years have passed since the world’s first burger made with cells grown outside of a cow was cooked and eaten at a news conference in London. The delicacy was presented by Mark Post, cofounder of Mosa Meat in the Netherlands, and was bankrolled by Google co-founder Sergey Brin to the tune of about £250,000.
Back in 2013, Post acknowledged that it would likely be another decade before such products would be available to consumers. Indeed, only one company – US-based Eat Just – is currently selling cultured chicken, albeit on a relatively small scale and just in Singapore.

Such products aren’t approved for sale in any other country, but scientists, investors and regulatory experts reckon that cultivated meat will hit the market in other nations like the US as soon as this year. They suggest the UK and Europe aren’t far behind and could be selling such goods in a year or two. Nations like Israel, Japan, China and Qatar also appear poised for regulatory approval in the near future.
Unlike plant-based meat alternatives, which use various additives mixed with plant protein to mimic the taste and texture of real meat, cell-cultured meat is produced using animal-derived cells, almost entirely decoupling meat from livestock. The starting point for traditional meat alternatives is a plant, from which a key protein is extracted. That protein isolate is then combined with other plant-based ingredients that act as binders, fats or flavourings to enhance the texture, taste, appearance and nutritional value of the fake meat.
In contrast, cultivated meat is made in a similar way to beer, but the process produces animal cells rather than alcohol. First, cells are taken from the tissue of an animal, via a simple biopsy. Next, some of those cells are screened and grown to make a bank of cells to be stored for later use. When the actual production process starts, cells from this bank are placed in a bioreactor and fed nutrients like growth factors, carbohydrates, amino acids, vitamins and minerals to help them grow and multiply.
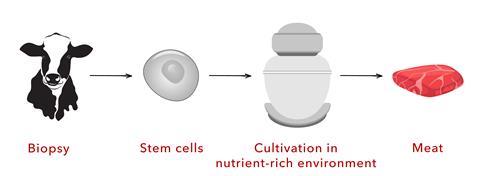
Once these cells have proliferated to billions or trillions, scientists change the nutrient conditions to trigger the cells to differentiate into various tissue types and assume characteristics of muscle, fat, or connective tissue. Finally, that tissue material can be harvested from the bioreactor and prepared via typical food processing and packaging methods. It is ‘meat’ without slaughter. Researchers are making fish and other seafood this way as well.
There are many different names for these products, including synthetic, lab-grown, cell-based and in vitro meat. This field of cellular agriculture has exploded as the cultivated meat industry swelled from about $50 million (£42 million) in 2018 to $366 million in 2020, according to analysis by Edison Research and the Good Food Institute (GFI). In 2021, cultivated meat companies raised $1.38 billion, which represented more than 70% of the all-time investment in this field, calculates GFI, a non-profit representing the alternative protein industry headquartered in Washington, DC.
There is no question that demand for meat worldwide will increase, and current production methods cannot satisfy that demand, so innovation in food technology is crucial. Mounting concerns about animal welfare and the need to mitigate climate change by decreasing greenhouse gas (GHG) emissions are adding fuel to the fire.
The explosion of cellular agriculture
By the end of 2021, 107 cultivated meat and seafood industry start-ups were announced – a 32% increase from the previous year, GFI says. There were only four such companies in 2016, according to ResearchAndMarkets.com.
Recent analysis by McKinsey determined that cultivated meat could be a $25 billion global industry and make up about 0.5% of the world’s meat supply by 2030. Edison went further, citing data from the global consulting management Kearney to predict that the field will see an annual growth rate of 41% to reach $600 billion by 2040, potentially outpacing the market for conventional and plant-based meats.
It takes about a month for the original stem cells to develop into mature fat tissue
Hoxton Farms, a London-based start-up dedicated to making animal fat, is among the companies that have emerged to eventually service future cultivated meat purveyors. Cofounded by synthetic biologist Max Jamilly and childhood friend and mathematician Ed Steele in 2020, Hoxton Farms takes animal cells, grows them in a bioreactor, and gives them instructions to become fat cells.
Under Hoxton Farms’ proprietary process, it takes about a month for the original stem cells to develop into mature fat tissue, which the company then collects from the bioreactor and sells to plant-based meat companies as a business-to-business ingredient. The intent is to provide for a truly authentic meat-eating experience.
‘They use it as an ingredient to make meat alternatives that look and cook and taste just as good as the real thing, if not even better,’ Jamilly tells Chemistry World. The plant-based fats that most meat substitute producers currently use, such as cocoa butter, coconut oil and palm oil, are not ideal. For example, they have undesirable melting points, and an unpleasant taste that often must be masked with salt or other additives.
Fat matters
Jamilly insists that fat is by far the most important sensory component of all meat. ‘It completely defines the way meat sizzles and browns and smells when you cook it,’ he says. ‘It defines the way that meat tastes as well – and that’s true whether you’re talking about high-end Kobe beef or simply the prosaic burger or chicken breast.’
The fat found in different species varies greatly
Hoxton Farms has figured out how to direct lipid biosynthesis to produce fats with various tastes, textures and nutritional profiles. This customisation is key to mimicking the many different types of animal fats found in the various cuts and types of traditional meats that exist. ‘Within a steak, for example, there’s the marbling that runs between the muscle, but then there’s also the fat that’s around the edge of the steak – and that’s just one kind of cut from one kind of breed,’ Jamilly explains.
There are significant distinctions between the fat in a cow from one part of the world versus another, and the fat found in different species also varies greatly. Pork and beef fat, for instance, taste completely unique.
‘We can make bespoke kinds of fat for specific applications and deliver them on demand,’ Jamilly says. ‘So, someone can tell us which kind of customised fat they need, and we can go away and with our scientific teams work out how to make it,’ he adds. However, Jamilly won’t share the details of Hoxton Farms’ proprietary processes, or the product lines that it offers.
In the long term, Hoxton Farms wants to sell to cultivated meat companies as well, but currently the planet’s sole supplier of cultured meat is Eat Just. That company was co-founded in 2011 by childhood friends Josh Tetrick and animal rights advocate Josh Balk in San Francisco, US, with the initial goal of finding plant-based alternatives to eggs.
From sci-fi to Singapore
Originally named Beyond Eggs, the company has gone through several names and is now known as Eat Just. It spawned a cultivated meat division called GOOD Meat in 2020, and has been selling its cultivated chicken products in Singapore since receiving approval from the Singapore Food Agency (SFA) in December 2020. ‘People think, very wrongly, that this idea of cultured meat is science fiction – that maybe it’ll happen 50 years from now,’ Tetrick says. ‘Meanwhile, consumers went to Mr. Loo’s “hawker” stand in Singapore just a few days ago and ordered chicken curry rice, sat down with their dad or their friends, and ate real meat without slaughter, and paid for it.’

The product can also be purchased at the JW Marriott Singapore South Beach’s Cantonese restaurant Madame Fan, and customers can even request home delivery.
Tetrick does not have a scientific background, but his company relies on a team of biochemists, protein chemists and culinary scientists. He is very open to using lots of different tools and ingredients, including plant proteins, yeast and animal stem cells lines. Jamilly agrees that the distinction between ‘plant-based’ and ‘cultivated’ likely will dissolve, and eventually all meat alternatives will be ‘hybrids’ combining plant-based and cultivated ingredients.
What’s in a name?
There is, however, an important semantic question about what can accurately be called ‘meat’. The ‘chicken’ product that Eat Just sells in Singapore is more than 70% material that came from animal cell lines, according to Tetrick. ‘It’s kind of an arbitrary line,’ he explains. ‘It was really just about maximising the flavour and texture.’
Legitimate questions are being raised about whether food can technically be described or labelled as ‘meat’ if it is derived only from an animal’s muscle cells, for example. Some argue that ‘muscle protein’ would be a more apt name.
There is also significant debate about whether, and under what conditions, cultured meat qualifies as kosher or halal, or even as vegan. Many suggest that the serum in which the initial animal cells were cultured and grown should be considered when answering such questions.
Regulatory restrictions represent one potential obstacle for cultivated meat. Such products will clearly attract more scrutiny everywhere as cellular agriculture becomes a mature industry and spreads across the globe.
If we receive approval, we’re going to be ready to go within weeks
In the US, for example, the Food and Drug Administration (FDA) and the United States Department of Agriculture (USDA) will have to approve any cultivated meat before it is available for consumption. The FDA will be responsible for overseeing activities at cell banks and facilities where cells are cultured, differentiated, and harvested to ensure the safety of these processes. Then, during the harvesting stage of the cell-culturing process, the USDA will assume oversight of production and labelling. This will include conducting inspections of the facilities where cells cultured from livestock and poultry are harvested, processed and packaged.
Tetrick says his company has had very fruitful interactions with the FDA and USDA, and that these agencies are approaching regulation thoughtfully. ‘If we’re fortunate to receive approval, we’re going to be ready to go within weeks,’ he says.
Meanwhile, a launch in Europe appears a bit further away, as the regulatory process there is more complex and time-consuming. ‘I think the estimate is that from the moment you submit a dossier it takes about a year and a half before you get approval,’ explains Leo Groenewegen, co-founder and chief executive of CellulaREvolution, a spinout from Newcastle University in the UK that is developing new ways to accelerate cultured meat production and make it cheaper.
Cutting the FBS
Beyond regulatory roadblocks, cell-based agriculture also faces technical obstacles, including the sky-high cost of the growth media that must be optimised for each cell line. Estimated to represent 99% of the production cost associated with cultivated meat, these are mostly only available in limited quantities that work just at small, lab-scale operations. In fact, the cost per litre for standard growth media has been ballparked at $377, which translates to $8600 per kilogram of final meat product .
Traditionally, cell culture media are supplemented with foetal bovine serum (FBS), which provides needed nutrients and growth factors. But FBS is extremely controversial because it is made from the blood of foetuses extracted from pregnant cows during slaughtering. Estimates indicate that just a litre of FBS can cost more than $1000. Besides being incredibly expensive, it fundamentally contradicts the whole concept of cruelty-free meat. Several companies however have been able to successfully develop serum-free cell culture media.
FBS is still used by most of the research labs
The serum that Eat Just uses to grow its cultured meat being sold in Singapore includes FBS – although it makes up less than 2% of the formulation, according to Tetrick. But the company has long been conducting R&D work that is serum-free and doesn’t rely on FBS. ‘We’ve submitted an application to the SFA with data showing why this serum-free formulation is optimal, and we’re waiting to hear back from them,’ Tetrick reports. The new process is under review and will hopefully be approved soon, he says.
But for now, FBS remains in use, and some say it is still the gold standard. In fact, it’s the only standard based on SFA’s approval of GOOD Meat. ‘It is still used by most of the research labs because it’s the easiest thing you can give the cells to grow them rather quickly for experiments,’ Groenewegen says.
Safety benefits
Cultured meat offers several significant benefits over conventional meat. For example, antibiotics are not fed to the cells, as they are to animals bred for human consumption, says Martina Miotto, the chief scientific officer and other co-founder of CellulaREvolution. ‘We actually know how much antibiotics are used, and sometimes abused, on real farms,’ she notes. The scientific literature provides evidence supporting an association between antibiotic use in food animals and antibiotic-resistant bacteria in humans.
Furthermore, it doesn’t appear that products harvested from animal cells can pass viruses to humans. This is important because concern about zoonotic diseases infecting people exploded after Covid-19 hit the planet about two-and-a-half years ago. The prevailing theory is that Sars-CoV-2 originated in bats, jumped to an intermediary animal at a so-called ‘wet market’ selling meat and seafood in Wuhan, China, and then was transmitted to humans.
‘Cultured meat will significantly reduce the risk of another similar pandemic,’ Miotto says, pointing to the markedly more sterile conditions in which these products are made when compared to slaughterhouses or wet markets. The cell-based food supply chain should contain no pathogenic bacteria or cross-contamination, she adds.
Eat Just’s Tetrick agrees. ‘These cells are free of salmonella, E. coli, faecal contamination, and there is little to no risk of zoonotic disease,’ he asserts. ‘It’s just an inherently more contained process than a chicken farm, for example.’
Cultured meat could even be made to be healthier than traditional meat by modifying, for instance, the amount of saturated fatty acids and polyunsaturated fatty acids in the final product. Unhealthy fats could potentially be replaced with healthier omega-3 fatty acids, for example, but researchers warn that this might mean a higher risk of rancidity that would need to be countered.
Climate concerns
But when it comes to the environmental impact of cultured meat, some question remains about exactly how much it will reduce GHG emissions, compared to conventional meat. The latest version of the Intergovernmental Panel on Climate Change report identified cultured meat as one emerging food technology that can bring a ‘substantial reduction in direct GHG emissions from food production’ by 2030.
Indeed, this new industry’s success would obviously mean considerably less land and water will be used to produce meat, compared to raising livestock. That avoids the significant emissions that have always been linked to rearing animals for food, but electricity is still needed to make cultivated meat.
I don’t think the environmental impact is smaller than for raising chickens right now
‘The culture conditions are very, very specific, so you basically have to maintain all of these bioreactors that use electricity and energy, and special culture media – very, very demanding culture media,’ explains Cindy Tian, an animal science professor at the University of Connecticut in the US. ‘To generate all of those materials needed to create the conditions for the cells to grow into chicken nuggets, for example, that’s not cheap – environmentally or otherwise.’
Tian is sceptical. ‘There is a lot of hope and hype for the lab-grown meat,’ she says. ‘I don’t think the environmental impact is smaller than for raising chickens or other animals right now.’ The process of making meat from animal cells remains ‘extremely inefficient’, Tian emphasises. Eventually, when the technology and infrastructure improve, the balance might tip, but she says right now cultivated meat is ‘very unsustainable’ and ‘there may not be a lot of good returns.’
In the meantime, however, important research continues. Tian and fellow University of Connecticut animal scientist Young Tang were recently able to induce bovine pluripotent stem (iPS) cells for the first time. These cells can potentially divide indefinitely in culture. The work, published in September 2021 and led by Tang, could have applications for cultivated beef by allowing scientists to grow entire cuts of meat from muscle stem cells differentiated from a single iPS cell.
The two researchers have joined colleagues in filing a provisional patent on the technology through their university’s technology commercialisation services. These bovine iPS cells could be used to develop germ cells in the lab that could establish in vitro breeding technology that is capable of producing generations of cows 10 times faster than traditional breeding techniques.
Going green
To try to gauge the environmental footprint of this field, GFI examined data from more than 15 companies involved in the cultivated meat supply chain last year, aiming to determine the future energy needs of a hypothetical large-scale ‘cultivated meat’ factory in 2030. If the systems were powered by renewable energy, then its carbon footprint was estimated to be around 90% lower than conventional beef, 52% lower than conventional pork, and 17% lower than conventional chicken.
Tetrick concurs that any cultivated meat production process must be fuelled by sustainable resources. ‘This field necessarily will need to be powered by renewable energy if we’re going to maximise our sustainability benefits,’ he tells Chemistry World. ‘You still need to power the bioreactor regardless of whether the inputs are more efficient – you’re still running a production process that requires energy.’
At scale, Tetrick insists that this emerging technology will be ‘unquestionably better for the environment,’ but he points out that any current calculation of such benefits is inherently limited. ‘We can attempt to predict what those energy requirements are at 250,000 litres, at 2.5 million litres, at 15 million pounds [6.8 million kg] per year, but they’re just estimates based on a model, and once we are actually producing cultivated meat at capacity we will have a much keener sense of the actual energy needs.’
The issue is more complicated than it might appear. Jean-François Hocquette, a research scientist at the French National Research Institute for Agriculture, Food and the Environment, says that although ruminant livestock produce GHGs like methane, their grazing has some environmental benefits. The presence of these animals stimulates the growth of vegetation because their manure affects the nitrogen cycle in a way that optimises microorganism activity.
‘They enrich the soil with a higher biodiversity of microbes, plants and more animals, which in turn provide more fertiliser for the soil,’ Hocquette explains. ‘If you decrease the number of farm animals because you can produce a lot of cultured meat, it’s important to consider the consequences because the virtuous cycle will be highly reduced,’ he warns.
We need to make tens of millions of pounds of cultivated meat
Meanwhile, several companies around the world are preparing to seek approval to sell cultivated meat by the end of this year, and many experts anticipate many such authorisations will happen before the end of 2023. These will likely be very small-scale launches in select boutique restaurants, or slightly larger food establishments.
There is general agreement that any full-scale commercial launch in which these products appear in supermarkets next to conventional meat and vegetarian options is still several years away.
Hypothetically, Eat Just could have regulatory approval from many more countries ‘tomorrow’, Tetrick says, but there would be a lack of infrastructure to supply enough meat to meet the demand. ‘The most important thing that we need to do is build a long-term capacity, both in the US and elsewhere – we need to make tens of millions of pounds of cultivated meat and then even more than that because the one thing we don’t have any uncertainty about is the demand,’ he says.
Conventional animal agriculture is inherently inefficient because it necessitates billions of live animals that all need to eat, which requires land to grow crops like corn, and the use of things like fertilisers. Currently, over a third of the planet’s usable land is dedicated to growing and sowing corn to feed the animals we eat, according to Tetrick.
If humans colonised a new planet, he suggests, even the chief executives of Earth’s largest meat companies probably would not propose to initiate conventional animal agriculture there. ‘It would be a bizarre suggestion because it only makes sense if it’s a habit that you’ve hooked into, that you feel like it’s necessary to continue in order to feed people,’ Tetrick says.
Rebecca Trager is the senior US correspondent for Chemistry World













No comments yet