Ten years of investment has turned nanotechnology into a booming research and industrial landscape. Fiona Case investigates whether it has lived up to the hype
In December 2010, politicians, funding agency representatives, venture capitalists and researchers involved with nanotechnology met in Washington, DC, to celebrate ten years of US National Nanotechnology Initiative (NNI) funding. The anniversary provides an occasion to reflect on achievements from a decade of nanotechnology research and development and a worldwide investment of more than $42 billion (£26 billion); and to consider the challenges for the future.

Interest in nanotechnology was growing in Europe during the late 1980s and 1990s: in 1986 the UK National Initiative on Nanotechnology was launched by the National Physical Laboratory and the Department of Trade and Industry; The European Union spent around €30 million (£26 million) a year on nanotechnology projects under the fourth framework programme (1994-1998). In the US, individual agencies established nanotechnology programs during the 1990s, ’but the investment was fragmented’, recalls Mihail Roco, senior advisor for nanotechnology at the US National Science Foundation (NSF).
In 1999 the NSF created a 10-year vision for nanotechnology. ’We had the opportunity to speak for 10 minutes at President Clinton’s Office of Science and Technology meeting,’ recalls Roco. ’We presented a vision of a technology that could detect and treat cancer, create light weight materials that were as strong as steel, and store the library of congress in the size of a sugar cube,’ he says. ’Clinton was intrigued,’ recalls Neal Lane, who was science and technology advisor to the president. The NNI became a top priority in his 1999 budget request. ’He called it his "tiny little initiative",’ recalls Lane.

But the initiative was not tiny, and it heralded the start of a decade of significant international funding for the field. By 2008 the US government was spending $5.1 (£3.2) per capita on research and development specifically for nanotechnology, Europeans were spending $4.6 per capita, the Koreans $6.0 and the Japanese $7.3. Global venture capital investment reached about $1.4 billion. ’The growth rate in international investment fell by more than half worldwide during the financial crisis in 2009,’ reports Roco, ’but appears to have returned to higher rates in 2010.’
What has this investment achieved? The number of researchers and workers involved in nanotechnology was estimated at about 400,000 in 2008; discoveries in nanotechnology were reflected in 65,000 Science Citation Index papers and 13,000 patents. Lux Research of Boston, US, estimates the value of products incorporating nanotechnology as a key component was about $254 billion in 2009. The market appears to be doubling every three years. ’In 2000 we made an estimate of a product value of $1 trillion by 2015,’ says Roco. ’Even with the financial crisis we are right on track,’ he says.
Coming of age
Although the numbers are impressive, as the nanotechnology community has met around the world to reflect on - even celebrate - the successes of the last decade, these figures have not been the focus. Excitement about revolutionary science and technology remains centre stage. Equally exciting is the success of an increasing number of companies bringing products based on nanotechnology into a range of different markets.
One of these burgeoning areas is energy sustainability, with nanostructured materials being developed for solar energy collection and conversion, energy efficient lighting, carbon capture and storage, and early work on artificial photosynthesis systems for creating fuels from sunlight. Boston, US-based firm A123 Systems have developed high-power lithium ion batteries based on nanoscale materials initially developed by Yet-Ming Chiang at the Massachusetts Institute of Technology (MIT) in Boston.

’NNI funding allowed us to scale up from the lab and improve the performance of the material, which now has become one of the leading technologies in the lithium ion battery industry,’ says Chiang
In a rechargeable lithium-ion battery the crystal structure of the cathode changes as lithium ions are added (battery discharging) or removed (recharging). The challenge is to engineer the material to allow that phase change to happen as quickly as possible and to prevent the material from phase separating as it cycles, since this will quickly limit the power of the battery. A123’s electrode materials are made up of particles less than 100nm in diameter, which helps overcome these problems (see Chemistry World, March 2010, p24).
’Conventional lithium batteries can be discharged in three minutes,’ says Chiang. ’The highest power batteries we developed, which were used in Formula One racing, can discharge 80 per cent of their energy in six to eight seconds.’ And the long term stability of the material? ’Some of the batteries we’ve been testing since 2006 are still going after 15,000 cycles,’ says Chiang.
A123 now has more than 2000 employees and is the leading manufacturer of lithium ion batteries for hybrid electric buses. But the technology goes well beyond just transport applications: ’A123 is a pioneer in the use of lithium ion batteries to help maintain a reliable and efficient power system,’ explains Chiang. ’The shipping-container-sized electric storage systems that we manufacture will enable greater use of renewable sources of energy such as wind and solar.’
Stronger, lighter, faster
Carbon nanotubes were early poster-children for nanotechnology. They promised new materials combining amazing strength with tunable electronic properties. Until relatively recently, many of these applications were held back by expensive and hard to control manufacturing methods.
But research into catalytic carbon vapor deposition in the laboratories of Janos B. Nagy at the University of Namur and Jean-Paul Pirard at the University of Liege, both in Belgium, led to techniques for creating and dispersing large quantities of high quality multi-walled carbon nanotubes (MWCNTs), and creation of spin-out company Nanocyl.
Nanocyl’s carbon nanotubes are now found in plastics, silicone, and epoxy-based resins for applications requiring electrical conductivity or protection from electrostatic discharge, enhanced mechanical strength, flame retardancy or thermal conductivity.
’Moving from laboratory based development activities to a small multinational organisation is undeniably one of the biggest challenges for a nanotechnology company,’ comments Monique Lempereur, Nanocyl’s global commercial executive director. ’It took us only five years to become the leading global manufacturer of speciality and industrial MWCNTs,’ she says.
The company moved from its pilot plant to industrial production in 2006, and inaugurated its first multi-tonne reactor - at Sambreville in Belgium - the following year. Nanocyl recently increased its production capacity of MWCNTs to more than 400 tonnes per year.
Keeping it safe

There are, however, some concerns that are shared across the entire community as we enter a second decade of nanotechnology. One of these is environmental health and safety (EHS), and particularly the effects of EHS legislation.
Governments and federal agencies worldwide agree on the need for significant efforts in EHS for nanotechnology. ’EHS was high on the agenda in Europe from the very beginning and is currently responsible for more than 5 per cent of EU investment in nanotechnology,’ says Christos Tokamanis , head of the nano and converging sciences and technologies unit at the European Commission. The US NNI has just issued a new EHS-strategy document, making it a primary objective for the next decade. There is, however concern that poorly designed legislation may limit development, and the uncertainty is definitely damaging.
A particular issue is the broad definition and regulation of nanotechnology based solely on the size of particles in a material. Pigments found in paints and varnishes have always contained nanoscale particles. Personal care products, soaps and detergents, and foods are naturally structured at the nanoscale - should they all be regulated and labelled?
Public concerns
’Everyone would agree that the safety question started out rather badly,’ says Kenneth Dawson, director of the Centre for Bio Nano Interactions at University College Dublin, Ireland, and coordinator of the EU infrastructure associated with nanosafety. ’There was an unfocused fear with very little basis in science,’ he adds. The public interest - even hype - that surrounded nanotechnology in the early days helped with funding initiatives, and inspired new generations of scientists. But it also led to scare stories about ’grey goo’. ’As a scientific community we were slow to respond,’ admits Dawson. ’Things have changed since then. But it’s like meeting a new person - it takes a long time to get over a poor first impression.’
’There is a genuine need to understand the interactions between nanoparticles and biology for EHS issues and beyond, says Dawson. ’You cannot control chemicals in the body,’ he explains, ’so you have to change their properties and restrict the dosage to avoid general toxicity. But our bodies talk to objects on the nanoscale. This provides the potential to send nanoparticles to specific places. If targeted therapeutics are going to work, it has to be on the nanoscale,’ he predicts.
One of the companies working towards this goal is US firm Cerulean Pharma, which is developing drugs containing nanoparticles to treat cancer. The company was founded in 2006 by Ram Sasisekahran from MIT, and Shiladitya Sengupta of Harvard Medical School, also in Boston. In addition, Cerulean has in-licensed technology developed by Mark Davis at the California Institute of Technology in Pasadena, US.
The physicochemical properties of Cerulean’s nanopharmaceuticals - such as size, surface charge and composition - affect their distribution in the body. They are designed to passively target the nanoparticles into certain tissues while reducing overall systemic toxicity. The nanoparticles remain within the bloodstream until they reach an area of ’leaky vasculature’ characteristic of cancer or inflammation, at which point they pass through the large pores of these blood vessels and release their drug payload.
’We need more effective and safer cancer treatments,’ says Sandra Glucksmann, senior vice president for research and business operations at Cerulean. ’People are now living longer and we want to be able to treat cancer patients for years without unpleasant side-effects.’
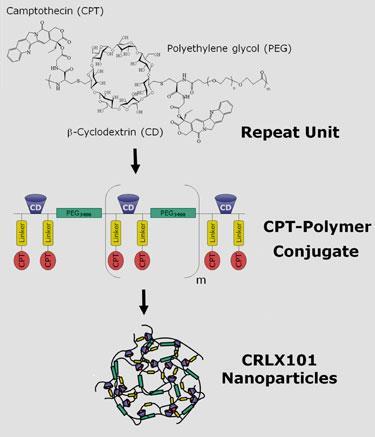
One of the Cerulean’s nanopharmaceuticals, CRLX101, is currently in Phase IIa clinical studies with advanced cancer patients. CRLX101 is a nanoparticle comprising the active molecule camptothecin, attached to a copolymer of poly(ethylene glycol) with a sugar-based cyclodextrin ring. ’During the early 1970s, camptothecin was evaluated in clinical trials, but failed because of its toxicity,’ explains Glucksmann. ’By incorporating camptothecin into targeting nanoparticles, CRLX101 is able to kill tumour cells while minimising the side effects typically associated with chemotherapy treatment,’ she says.
The potential for nanotechnology in medicine goes beyond drug and gene delivery. At the NNI Summit, Jeff Schloss, program director at the US National Institutes of Health (NIH), talked about the NIH nanomedicine initiative. ’We are learning how cellular machines operate at the nanoscale level and then using these design principles to manipulate and re-engineer biology,’ Schloss explains. ’For example, nanomaterials are being studied as self-assembling scaffolds for bone repair and neural tissue regeneration,’ he says. ’Nanopore devices are being investigated for single-molecule sequencing of DNA without fluorescent labelling. This could dramatically reduce the cost of genome sequencing and individualised patient treatment.’
People power
Schloss admits that training nanomedicine researchers remains a challenge. ’We need multidisciplinary people who understand both nanotechnology and medicine,’ he says. He hopes that new NIH Nanomedicine Development Centers will not only produce research results, but also people to form new companies. ’Large companies are rather conservative with respect to nanotechnology,’ he points out. ’Most commercialisation is happening through small start-ups.’

Jeremy Baumberg, director of the Nano Doctoral Training Centre at the University of Cambridge, UK, agrees. ’Nanoscience and nanotechnology are areas in which students need a broader range of skills than more traditional research fields,’ he says. Cambridge is one of several universities across the world aiming to train multi-discipline nanotechnologists for the 21st century. For the first year, PhD students do not know what they will research, or indeed in which department. They do practical work in every conceivable place - from transmission electron microscopy to growing nanoparticles, making devices and cloning DNA - and take lecture courses across many disciplines. ’We take them completely outside their comfort zones. We also stress innovation during a range of courses in high-tech entrepreneurship,’ he explains.
’I would have loved to do a PhD like this,’ enthuses Baumberg. ’This is one of the most important things we can do within UK science, but it is expensive. The Engineering and Physical Sciences Research Council has put in more than £200 million at a very difficult time, they ought to be applauded for the initiative,’ he adds.
Looking ahead
So, as the US NNI tenth anniversary celebration concludes, and the world continues to reel from the financial crisis in 2009, what are the prospects for continued growth and funding of nanotechnology research?
’The climate in which science and technology investment will be considered in the US has become more difficult,’ says Raymond Garant, director of public policy at the American Chemical Society. ’Funding for even the most worthy programmes is being measured in light of the consensus that the nation’s debt must be dramatically reduced.’ But there is some hope. ’The bipartisan support for the America Competes Act reauthorisation that became law in early January is a good sign that there are still leaders committed to investment in science and technology innovation,’ he adds.
Mihail Roco is hopeful. ’We have strong support in Congress. They see nanotechnology as one possibility for maintaining high paying jobs in the US. Even if other fields are subject to cuts, I don’t think nanotechnology funding will be cut in the current budget,’ he says. The NNI has ambitious plans for the next decade including signature initiatives in nanoelectronics, nanomanufacturing and nanotechnology for solar energy collection and conversion.
At the EC, Tokamanis is somewhat concerned. ’Government investment in nanotechnology has peaked. It’s not going to grow any further because of competition from other priorities,’ he predicts. ’However, two thirds of European investment in nanotechnology R&D currently comes from government, only one third from industry. The most important thing we can do is to reduce the barriers to industrial investment.’
’We need Europe to stay focused, and not be distracted or discouraged by the safety issues,’ says Dawson. ’Europeans are more cautious about these questions in all arenas - but they could really pioneer the introduction of new technology, safely,’ he says. ’If we simply stand back, others will introduce these technologies for us. We will end up buying them in our goods, and have no influence as to how things are done.’
Fiona Case is a science writer based in Vermont, US.
Additional information
- Nanotechnology Research Directions for Societal Needs in 2020 (M Roco, C Mirkin and M Hersam, eds.), Springer 2010
- Nanotechnology at the European Commission
- Research Councils UK Nanoscience Programme












No comments yet