This riddle has come to vex certain corners of the catalysis community. But once solved, it could potentially point to new kinds of chemistry, as James Mitchell Crow discovers
The story typically starts with a known catalytic reaction - a carbon-carbon bond forming cross-coupling reaction, say - that relies on a noble metal catalyst such as palladium or platinum. With the growing awareness that such metals are a finite resource, as well as expensive, seeking ways to carry out the same key reactions with a more abundant metal such as copper or iron - or even ditching the metal altogether - has become a hot topic of research.

In many cases, alternative catalysts to noble metals have been successfully found. But a spate of recent examples present a cautionary tale that all might not necessarily be as it first appears - that a trace impurity in the reaction mixture might be the catalytically active species. And even experienced chemists can be caught out.
’I’m not so proud about this area,’ says Carsten Bolm, who is renowned for his work on enantioselective catalysis at RWTH Aachen University in Germany. In the late 2000s, Bolm and his team published a series of papers reporting iron-catalysed cross-coupling reactions. ’We looked into a reaction that worked with palladium. People had gone from palladium to copper and it was known to work with copper, and we wanted to simplify it even further so we looked into iron.’
However, Bolm was contacted first by Stephen Buchwald at the Massachusetts Institute of Technology in Cambridge, US, and then by Per-Ola Norrby at the University of Gothenburg in Sweden. Both suggested that iron wasn’t responsible for the observed reactivity, but rather that trace quantities of copper were the catalyst. ’We had an almost year-long debate about this, because we could not believe that such small quantities of copper would have that impact,’ says Bolm.
Subsequent studies did show that minute amounts of copper, an impurity in the iron chloride that Bolm was using - equivalent to just a few parts per million in the reaction mixture - were responsible for catalysing the observed cross-coupling reactions.1,2
Scratching the surface
It’s not the only example. One of the earliest of the recent series of cases came from Nicholas Leadbeater’s lab at the University of Connecticut in Storrs, US. In the early 2000s, Leadbeater was investigating the use of microwaves to promote Suzuki cross-coupling reactions. In some cases, the reaction worked well even when no palladium catalyst was added into the reaction mixture.
In a bid to rule out the presence of any rogue metal, the team used new glassware, new reagents and analysed the crude reaction mixture for any trace of palladium. None was found, down to the 0.1ppm detection level of the analytical apparatus used. The team published the paper under the title Transition-metal-free Suzuki-type coupling reactions.3
As Leadbeater later discovered, the reaction mixture did contain palladium after all. Just 50ppb was enough to promote the reaction, and the source of the contaminant was the sodium carbonate base that they were using.4
So what is behind this apparent spate of recent cases? ’I think that this is an age old issue, ’ says Buchwald. ’The problem lately is that people are making a big deal out of using new metal systems, so the fact that it is the old metal that is probably doing the chemistry is more highlighted. ’
However, the string of cases certainly has its upsides - not least in that it sends a clear cautionary message to the catalyst community. ’What I’m kind of happy about is that these papers by Buchwald, Norrby and myself really stimulated the community to look into this issue a little bit more,’ says Bolm. ’If you read the papers now on iron catalysis, they refer to these two papers, and list the studies that they did to make sure that this is iron catalysis. The community sees the difficulties now, particularly with iron.’
The message certainly seems to be getting out, and not just for iron. A similar problem recently struck Zolt?n Nov?k and his group at E?tv?s University in Budapest, Hungary.5 Nov?k was investigating the Sonogashira cross-coupling reaction, which classically involves a palladium-copper catalyst mixture. The team wanted to know whether copper catalysts could be found that didn’t need the more expensive palladium co-catalyst. Initially they had great results with their new copper complexes, getting complete conversion within 6 hours at just 50?C. However, aware of cases such as that of Leadbeater, the team became suspicious of the bottle of caesium carbonate that they were using as a base. ’We bought new caesium carbonate, and the reaction stopped working so well,’ says Nov?k.
Marvellous nanomaterials
Are carbon nanomaterials such as graphene and carbon nanotubes all they’re cracked up to be? Or might impurities be responsible for some of the remarkable properties often attributed to them?
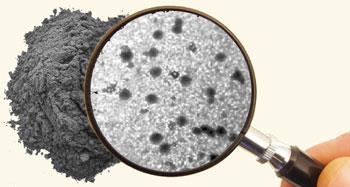
For some examples, that is certainly the case, says Martin Pumera, who researches the electrochemistry of nanomaterials at Nanyang Technological University in Singapore. ’Carbon nanotubes are highly heterogenous materials - they are grown from catalytic nanoparticles, which are usually metallic, and after the growth these catalysts are practically impossible to remove.’
That might not matter for applications such as composite materials, where the high strength of the carbon nanotubes is the key. But for applications such as electrochemistry, these metallic nanoparticles, trapped within the tubes, can have a huge impact, says Pumera.
The first person to draw attention to this issue was Richard Compton at the University of Oxford, UK, who in 2006 showed that the electrocatalytic properties attributed to certain nanotube-modified electrodes were actually due to metal impurities rather than the nanotubes themselves.6
’Seeing that, I couldn’t resist following up the work,’ says Pumera, whose team started to look for other impurities that might affect the nanotubes’ electrocatalytic properties. ’I have a paper that shows the electrochemistry of some carbon nanotubes is influenced by impurities within impurities,’ he adds. The nanotubes were contaminated with residual catalytic nanoparticles of cobalt, which were themselves contaminated with iron - and it was the iron that was responsible for the observed electrocatalysis.’ If you look, some of the impurities contain about 10-100 atoms of iron. Those 10 or 100 atoms are responsible for the huge catalytic effect.’7
A question of characterisation
It’s not just metals that contaminate samples of carbon nanotubes, either. They also invariably contain some amorphous nanographitic carbon, which is also electrocatalytically active. Pumera says that he is concerned that people continuing to attribute such properties to the nanotubes themselves risk damaging the whole field, as industry will become wary of using nanotubes if they find that the reported results don’t hold up. ’We just want to clear the air and say, ok, maybe if the nanographitic impurities are responsible for the catalytic properties, why bother with carbon nanotubes at all, why not just use nanographite? The same with the metal nanoparticles.’
However, that’s not to say that nanotubes can’t be useful for catalysis - it’s just that their role should be properly described, says Pumera. ’Carbon nanotubes are beautiful nanowires, so to have the metallic nanoparticles as the catalytic centres and the carbon nanotubes as nanowires - I believe that this is something that we should be doing.’
|
’I certainly agree that the role of impurities on the behaviour of carbon nanotubes is still neglected by many authors despite clear messages in the literature,’ says Compton. ’I would not be as pessimistic, however, regarding the damage. It’s probably more important to look at progress at the leading edge rather than worry about the reverse,’ he adds. ’After all, my 2006 paper which first pointed out this issue already has over 150 citations, so people are taking notice!’
|
And on the flip side, Pumera has also begun to show that the toxicity sometimes ascribed to nanomaterials can be due to impurities rather than the nanomaterials themselves.
Metal-free myth?
Having recognised the potential problem when looking for new metal catalysts, what can be done about it? First of all, it’s important to keep things in perspective, says Bolm. Despite the recent string of cases where metal impurities are the catalytically active species, what is certainly clear is that only a tiny percentage of the catalytic reactions reported in the literature have been affected in this way. ’It is clear to me that this is a very special condition that we used,’ says Bolm. ’It is just a small number of reactions that are affected, and we were fortunate - or unfortunate - enough to stumble across one, using conditions where copper is so extremely active.’
As the latest analytical techniques reveal, dealing with the issue by removing all the unwanted metal in a reaction mixture isn’t an option. ’With a good analytical partner who uses ICPMS [inductively coupled plasma mass spectrometry], we can measure transition metal contaminants down to ppb, or even ppt levels,’ says Nov?k.
Bolm agrees. ’The question to me is, can you really ever claim that a reaction is metal-free? Because if you look hard enough, we have techniques now such as atom absorption spectroscopy that find such small quantities that essentially no reaction is metal-free anymore.’
So it might be impossible to remove all of the impurities from a reaction - but if the reaction works, does it really matter if it is an impurity that is key to that success? Could we knowingly rely on impurities to drive reactions? The approach might be intellectually unsatisfying, but is it a pragmatic approach that industry could apply for molecule making?
In fact, industry struggles with this problem just as much as academia does, says Nov?k. The trouble with that approach is that reagents vary in their contamination profile from batch to batch and from vendor to vendor. A reaction might work well on a small scale in a discovery lab, but that’s no guarantee of success further on in the process. ’The reaction doesn’t work because the contamination profile is different.’
Spotting the culprit
All this seems to leave us back at square one: needing to work out which reactions have been influenced by impurities. So what can be done? Picking out which reactions might be susceptible to interference from impurities is difficult. But when trying out a new catalyst on an old reaction, there are clues in the patterns of reactivity that can reveal whether trace metal impurities are having an impact or not. ’The red flag I always look for is whether the "new" chemistry is general, or whether it only works for the simplest examples of the old chemistry,’ says Buchwald.
|
’[In the Bolm case] we questioned why the same ligand was used for iron and copper,’ Buchwald adds. ’We also felt that since the reactions were so much slower with the iron systems that they might be due to trace copper. ’
|
The other key is to look at whether the new metal catalyst seems to work in just the same way as the old metal - whether it works well on the same kinds of substrates. ’If you find totally different reactivity then I think you can safely say that it has something to do with the metal that you put in, and not an impurity,’ says Bolm. ’So I think people should look for different reactivity.’ They should also try out different sources and purities of the metal to see whether that has any unexpected impact on reactivity, he adds. ’And finally just report what source you used. In all the papers that we have we wrote down the source and its purity - you can repeat our experiments, which is why we never retracted our paper.

’We describe our chemistry now describing the limits that we found,’ Bolm adds. ’So we say, if this ppb amount of palladium has any impact, we cannot change it. We use new flasks, and clean everything as much as we can, but if we look hard enough we certainly find metal. It’s a very weird situation.’
However, as well as an increased awareness of the issue, there might be one truly positive outcome to these discoveries of catalysts that are highly active - they might eventually point the way to new kinds of chemistry in which minimal amounts of catalyst are required.
’It is still a question to me why the copper is so active in ppm quantities,’ says Bolm. ’You can wonder if the ligand in excess forms some clusters of the right size that are active or something. So this could be a direction of research that stems from the finding that an impurity plays a role.
’What I think will also be a question in the future is co-catalysis. There are cases that you have co-catalysis between the copper and the iron, or many other metals. I can see that something like co-catalysis would be stimulated by these impurity discussions, but I don’t see it at the present time.’
At the very least, it’s a good reminder to the community to always try your reaction with very low levels of catalyst, to see whether it will still work, says Duncan Macquarrie, a member of the Green Chemistry Group at the University of York. ’It’s certainly worth trying deliberately,’ he says. ’There are examples, particularly for polymer production, where you can use tiny quantities of catalyst to make huge amounts of product. ’
James Mitchell Crow is a freelance science writer based in Melbourne, Australia
References
1 S L Buchwald and C Bolm,Angew. Chem. Int. Ed., 2009, 48, 5586 (DOI: 10.1002/anie.200902237)
2 P-F Larsson et al, Angew. Chem. Int. Ed., 2009, 48, 5691 (DOI: 10.1002/anie.200902236)
3 N E Leadbeater and M Marco, Angew. Chem. Int. Ed., 2003, 42, 1407 (DOI: 10.1002/anie.200390362)
4 R K Arvela et al, J. Org. Chem. 2005, 70, 161 (DOI: 10.1021/jo048531j)
5 Z Gonda, G L Tolnai and Z Novák, Chem. Eur. J., 2010, 16, 11822 (DOI: 10.1002/chem.201001880)
6 C E Banks et al, Angew. Chem. Int. Ed., 2006, 45, 2533 (DOI: 10.1002/anie.200600033)
7 M Pumera and H Iwai, Chem. Asian J., 2009, 4, 554 (DOI: 10.1002/asia.200800420)












No comments yet