Ruthenium complex provides environmentally friendly route to key industrial intermediates
Researchers based in Israel have developed an easier way to make troublesome primary amines, using a new ruthenium catalyst.
As David Milstein, of the Weizmann Institute, Rehovot, explains, compounds ending in amine groups (-NH2) are routinely used in the manufacture of industrial chemicals and pharmaceuticals, but making terminal primary amines selectively is difficult.
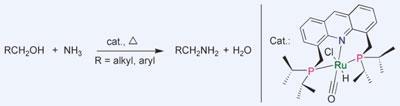
On a commercial scale, amines are made by reacting alcohols and ammonia together at high temperatures and pressures. But the initial primary amine products are extremely reactive and tend to react further with alcohols to form secondary or tertiary amines under the reaction conditions. Any surviving primary amines must be picked out of the mess afterwards.
Milstein and colleague Chidambaram Gunanathan have now developed a low temperature, low pressure approach to amine synthesis, avoiding harsh conditions but giving high yields of primary amines. ’The selective catalytic synthesis of primary amines directly from alcohols and ammonia with elimination of water, under relatively mild conditions, without producing waste, is highly desirable both economically and environmentally,’ says Milstein.
The key to the reaction is the team’s catalyst, a complex of ruthenium (II) pincered inside a tridentate ligand. Exactly why the reaction works so well is still unclear, but the researchers suggest the reaction could go via the formation of an aldehyde that reacts with ammonia to form a hemiaminal intermediate. This eliminates water to form a terminal imine, which is then reduced to the primary amine product by the catalyst. Milstein says he has already had inquiries from industry and has applied to patent the reaction.
’This paper is another very impressive stepping stone in the catalytic properties of ruthenium pincer complexes from the Milstein group,’ says Walter Leitner, an expert in industrial chemistry and catalysis at RWTH Aachen University, Germany. ’This new catalytic reaction is formally a mono-alkylation of ammonia by a primary amine - a ’dream reaction’ indeed.’
Susan Aldridge
Enjoy this story? Spread the word using the ’tools’ menu on the left.
References
C Gunanathan C and D Milstein, Angewandte Chemie, 2008, 47, 8661 (DOI: 10.1002/anie.200803229)






No comments yet