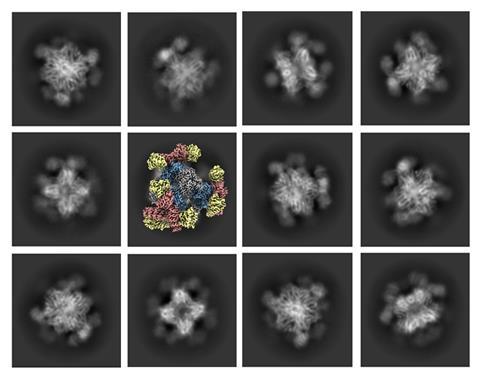
Cryo-electron microscopy can now determine the atomic structure of small and medium-sized proteins thanks to molecular scaffolds that hold the proteins in place. The work could assist the discovery and design of promising drugs by elucidating protein structures that were previously inaccessible by cryo-EM.
Cryo-EM scooped the Nobel prize in chemistry in 2017 as a revolutionary technology that images the atomic structure of biological molecules in high resolution. It works by firing a beam of electrons through a frozen sample to produce thousands of photographs of a molecule’s orientations. A computer then processes these images to produce a composite, high-resolution 3D image of a desired molecule.
However, the technique was limited to imaging relatively large biomolecules with molecular masses above 50kDa. Anything smaller made it difficult to clearly determine a molecule’s orientation, which resulted in blurry or low-resolution images. Researchers have previously tried a workaround by binding small proteins of interest to a larger scaffold in order to make the overall molecule large enough for cryo-EM to image clearly. However, these attempts fell short because the binding wasn’t sufficiently rigid to accurately reconstruct a high-resolution image.
Now, Roger Castells-Graells and his colleagues in Todd Yeates’ lab at the University of Californian, Los Angeles (UCLA) in the US, have found a solution by designing a cube-shaped protein scaffold that has tripod-like protrusions, which firmly bind smaller proteins in place in order to image them.
‘Attaching the small molecules rigidly to larger scaffolds creates particles that are large enough to be imaged, and which all have precisely the same 3D shape,’ Yeates says. ‘From there, the process works as usual to construct the high-resolution 3D image.’ While the scaffold is imaged too, this can be digitally removed, leaving just the structure of the small protein target on the 3D image.
To test the approach, the team set out to image mutants of a small protein just 19kDa in size called Kras, which encourages cells to proliferate and is mutated in about 25% of human cancers. The researchers found that their scaffold enabled them to image Kras, which matched known structures previously determined with x-ray crystallography. Further experiments showed how a Kras mutant binds to a potential lung cancer drug. Results revealed new insight into a structure conformation that differed from reported x-ray structures.
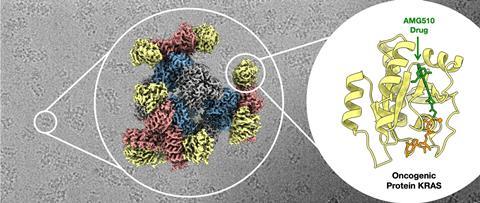
‘This is a very elegant solution to the issue of size limitation in the use of cryo-EM,’ comments Paul Race, a biochemist at Newcastle University, UK, who regularly uses cryo-EM. ‘The exciting development here is in the generality of the approach – the scaffold can be reconfigured to bind a multitude of different proteins. In principle, this could provide access to a raft of new protein structures, but there is a need to test the approach with as broad a range of candidate proteins as possible,’ he adds.
UCLA has now filed a patent on the new technique. Meanwhile, Yeates, Castells-Graells and colleagues have founded a start-up company called AvimerBio with a view to develop commercial applications in collaboration with the pharmaceutical industry.
References
R Castells-Graells et al, Proc. Nat. Acad. Sci. USA, 2023. DOI: 10.1073/pnas.2305494120









No comments yet