Bioimaging probe excited by longer wavelengths of light but doesn’t suffer from long-term retention in the immune system
Scientists have designed a safe fluorescent probe that can be coupled to a protein for accurately imaging tumours deep in the body. The probe could guide surgeons performing surgery.
Visualising tumours deep in the body is vital for accurately diagnosing and treating cancer. Bioimaging probes excited by conventional near-infrared light (700–900nm) are hindered by their inability to penetrate many cell layers. So-called NIR-II probes, fluorescing in the 1000–1700nm region, however typically suffer from high uptake by healthy tissues, making imaging unclear, and long-term retention in the body, which can be risky for patients.
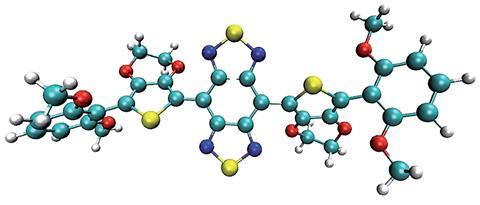
Now, Xiaoyuan Chen, from the National Institute of Biomedical Imaging and Bioengineering, a part of the National Institutes of Health, US, and colleagues have developed a NIR-II probe that is rapidly excreted renally, with minimal uptake by the liver and immune system. The probe has a shielding–donor–acceptor–donor–shielding structure; the acceptors units comprise benzothiadiazole structures, and 3,4-ethylenedioxythiophene substituted thiophene acts as the bridging donor unit. It has a higher quantum yield than other NIR-II probes thanks to dialkoxy-substituted benzene units that shield the fluorophore from intermolecular interactions; allowing imaging with a lower patient dose and a shorter laser exposure time.
By conjugating the probe with TATE, a peptide that has receptors on neuroendocrine tumours, and injecting it into tumour-bearing mice, the fluorophore–peptide conjugate had a tumour-to-normal tissue ratio of ~10 – commonly used positron-emission tomography provides a ratio of 6. Such level of detail allowed the team to excise a tumour using white light as a guide. Coupling the fluorophore with other peptides allowed it to visualise deep brain tumours through the skin and scalp.
Chen says ‘the present conjugates demonstrate renal excretion behaviour with minimal toxicities and low organ uptake, performing as arguably the brightest targeting NIR-II conjugates with excretion ability ever reported’.
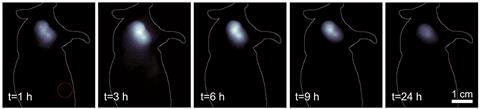
The team found that the probe not only has to be less than 5nm wide for renal excretion, but it also has to have low affinity for binding to plasma proteins and a low macrophage uptake. Mice renally excreted the conjugate within 6–12 hours.
Xuechuan Hong, an expert in molecular imaging at Wuhan University, China, says ‘this is much safer for humans because it can be timely excreted through urine. Such new molecular imaging could improve glioblastoma brain, gastroenteropancreatic neuroendocrine, liver-cancer detection and NIR-II image-guided tumour surgery.’














No comments yet