5f and 6d orbitals in low-valent actinides exhibit unprecedented charge transfer in actinide–metal complexes
Scientists have made actinide–metal complexes where actinide ions act as electron donors for the first time.
One of the key features of the early actinides is that their 5f orbitals participate in bonding. However, there is little evidence of this for thorium.
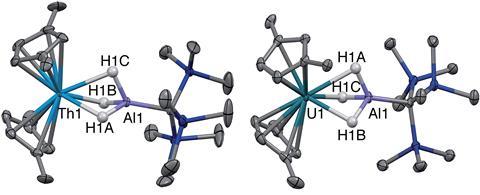
The team, led by John Arnold from the Lawrence Berkeley National Laboratory in California in the US, wanted to make a low-valent thorium–metal complex to gain insight into thorium’s electronic structure. They created the thorium(III)–aluminium complex [Cp‡2Th(μ-H)3AlC(SiMe3)3] and its uranium(III) analogue and analysed their electronic structures using electron paramagnetic resonance (EPR) spectroscopy and a variety of computational techniques.
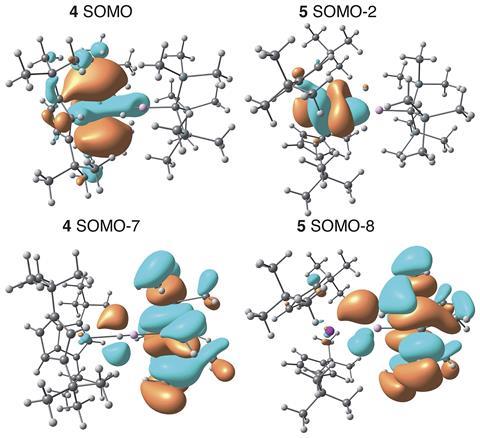
The EPR hyperfine interactions between the single thorium(III) electron and the aluminium atom indicated charge transfer from the thorium metal centre to the aluminium one about three times greater than that of the titanium(III) analogue. This remarkable result was repeated for the uranium(III)–aluminium complex and shows the reverse of the typical donor–acceptor paradigm for actinide–metal bonds with the charge moving away from thorium and uranium towards aluminium. Natural bond orbital analyses confirmed donation from the occupied thorium 6d and uranium 5f orbitals to the aluminium 3p orbital.
These complexes are the first examples of actinide–metal dative interactions. The next steps for Arnold’s team are to quantify the nature of the aluminium interactions in this system and to expand the chemistry of low-valent thorium with other Lewis acids, which they hope will lead to more insights into actinide metal bonding.

![An image showing the molecular structure of the [FeFe]-hydrogenase active site, the H-cluster](https://d2cbg94ubxgsnp.cloudfront.net/Pictures/380x253/1/9/3/501193_index_834443.jpg)









No comments yet