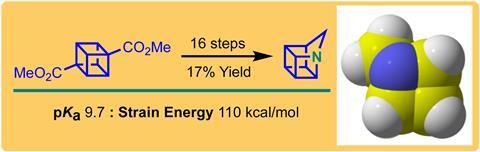
Researchers in Australia and the US have synthesised and characterised 1-azahomocubane,1 over 50 years after the first reported synthesis of cubane.2 Their 16-step synthesis overcomes the main bugbear of making cubane frameworks – undesired rearrangement reactions of high-energy intermediates.
Cubanes are a family of molecules in which eight carbon atoms form the vertices of a cubic framework. The tight bond angles in these frameworks create significant strain energy in the molecules. Homocubanes are homologues of cubanes in which one edge of the cube contains an extra carbon atom, for a total of nine carbon atoms. Azahomocubanes exchange one carbon atom of a homocubane for a nitrogen atom and have been unreported, until now.
Craig Williams at the University of Queensland, Philip Eaton at the University of Chicago, and their co-workers synthesised 1-azahomocubane by using an aza-variant of the Favorskii reaction to form the strained ring system. They were also able to crystallise 1-azahomocubane as a 2:1 complex with silver perchlorate. Williams says they were ‘extremely lucky’ to obtain the crystal structure after one of his co-workers ‘went back to the fridge about eight months later and discovered beautiful crystals inside the [NMR] tube.’
‘It’s chemistry that deals with very fundamental platonic hydrocarbons,’ comments Mathias Senge, an organic chemist at Trinity College Dublin in Ireland. ‘It’s not something that can be done that easily despite all of the new fantastic reactions which have come out in the past 20 years. You have to somehow deal with the strain in the molecules. There are not many people, and Craig Williams is one of them, who can use strain as a guiding principle in synthetic development.’
Susannah Coote, an expert in synthetic photochemistry at the University of Bath in the UK, also praises the researchers’ ingenuity. ‘The aza-Favorskii-type step is certainly very impressive. It’s a great example of how creativity is so important in organic chemistry – initially it’s difficult to see how you could go from the starting material to the product in so few steps, but it works!’
1-azahomocubane was surprisingly stable as the free amine, but Senge says this is more common than you might expect for such seemingly strained structures. ‘You have 90° angles with sp3 hybridised carbons. What happens? Should this thing go boom immediately? No, actually, it doesn’t.’ 1-azahomocubane was also over 10 times less basic (pKa = 9.7 ± 0.1) than other nitrogen-containing frameworks. ‘That was really a little bit surprising,’ adds Senge.
Finally, Senge comments that much about cubanes remains to be studied: ‘Many people call them scaffolds but they don’t use them as scaffolds. Using them as scaffolds – that’s where the next step will lie.’ ‘More work is definitely needed to develop synthetic routes to derivatives [of 1-azahomocubane] to provide enough material for evaluation’ adds Coote. Williams, himself, is optimistic: ‘I think there’s a bright future for azalogues of fundamental hydrocarbons. Having a nitrogen atom present within the skeleton definitely enhances its attractiveness.’
References
1 T Fahrenhorst et al, Chem. Sci., 2023, DOI: 10.1039/d3sc00001j (This article is open access.)
2 P E Eaton and T W Cole, J. Am. Chem. Soc., 1964, 86, 3157 (DOI: 10.1021/ja01069a041)








No comments yet