A photocatalytic system has been created that can turn carbon dioxide into basic chemical building blocks
Japanese researchers have shown that photocatalytic reduction of CO2 is possible using water as the reducing agent. The work boosts the possibility of a clean route to use CO2 as a feedstock to produce environmentally friendly chemicals and solar fuels.
At present, most methods of converting CO2 to chemicals need a lot of energy because CO2 is very stable and inert. Photocatalysts have shown promise as an alternative, however it remains a challenge to develop a highly reactive and efficient catalytic system required to activate the molecule.
Moreover, for a system to be economically and environmentally viable at the industrial scale, it must be based on inexpensive and abundant metals and use renewable energy. Previous research has hinted that water might provide a clean way to provide the electrons necessary for CO2 reduction, but no one could confirm it.
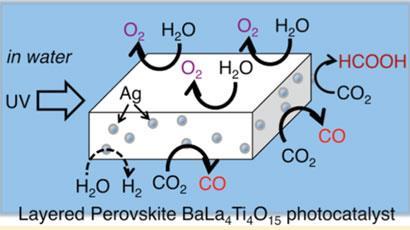
Now, Akihiko Kudo and colleagues at the Tokyo University of Science have demonstrated a photocatalytic CO2 reduction process in which water can be used as the electron donor and hydrogen source. ’We need only water, light and a photocatalyst. No other chemicals are required for the CO2 reduction in our system,’ says Kudo.
The team conducted experiments with several metal oxide photocatalysts known to be efficient at splitting water. These were loaded with different co-catalysts in order to create a suitable reaction site for CO2. The most successful of these was a silver loaded BaLa4Ti4O15 photocatalyst powder which was dispersed in water that had CO2 bubbled through it. When this system was illuminated with UV light at room temperature and pressure carbon monoxide was produced - a key compound for the chemical industry. CO2 can also react with hydrogen split from water to produce formic acid (HCOOH).
’The confirmation of water as the electron donor enables the design of a photocatalysis system by coupling the water oxidation half-reaction with the CO2 reduction half-reaction to produce CO and hydrocarbons without the need to use a sacrificial reagent (electron donor),’ says Huijun Zhao, a photocatalyst expert at Griffith University in Queensland, Australia. ’This is a step closer to realising an ideal solar fuel production system that utilises abundant water and CO2 as the reactants to produce useful chemical fuels.’
Kudo is keen to emphasise that this work is still very much at the fundamental level; however, he is hopeful that it could have practical value. ’It is important from a viewpoint of environmental and energy issues to utilise CO2 as a carbon source,’ he says. ’We will be able to produce fuels and chemicals by photocatalytic CO2 reduction if we can find efficient photocatalysts. If the practical use is achieved, consumption of fossil fuels can be suppressed.’
’When dealing with CO2 reduction, the devil is in the detail and one of the most challenging aspects is the design of systems able to address all the constraints of the large scale recycling of a very stable chemical waste,’ says Thibault Cantat from the French Alternative Energies and Atomic Energy Commission. ’These results are both promising and exciting because they succeeded in this difficult task by developing a silver photocatalyst (Ag/BaLa4Ti4O15) and can utilise UV light, as an energy source, and water, as an abundant electron source, for the reduction of CO2 into useful carbon monoxide.’
Kudo’s team is now looking to improve the system and use visible light instead of UV radiation to trigger the process. ’We are trying to develop highly efficient photocatalyst systems that work using solar radiation and to produce not only CO but also methanol and methane,’ adds Kudo.
James Urquhart
References
et al, J. Am. Chem. Soc., 2011, DOI: 10.1021/ja207586e






No comments yet