Infinity shaped molecule, infinitene, is aromatic, according to calculations done by scientists in Finland. The finding opposes widely held rules on aromacity.1
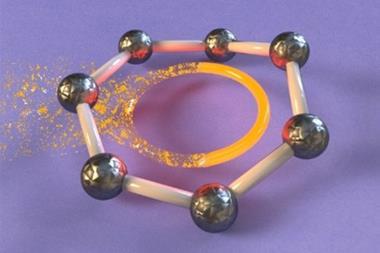
Newly synthesised this year by researchers at Nagoya University, Japan, infinitene is a looped polyarene assembled by fusing 12 benzene rings.2 This new form of nanocarbon adds to a family of non-planar aromatic systems with fascinating twisted or looped topologies and creates new opportunities for extending the boundaries of aromaticity.
Understanding the electron delocalisation behaviour in non-planar systems is challenging. While the Hückel rules are commonly used for planar systems, the aromaticity of twisted molecules can typically be understood using the Möbius rules. They relate the number of twists and π-electrons in a conjugated structure to its aromaticity. According to the Möbius rules, infinitene should be antiaromatic.
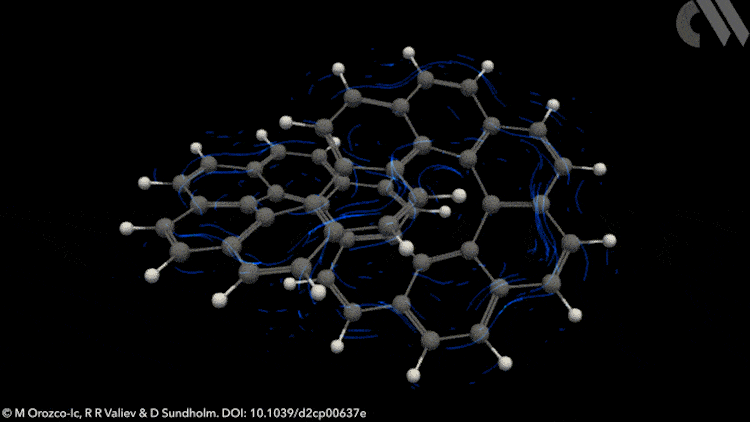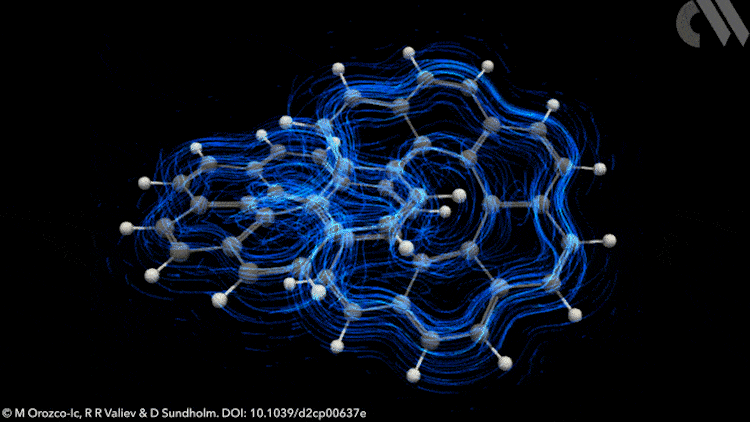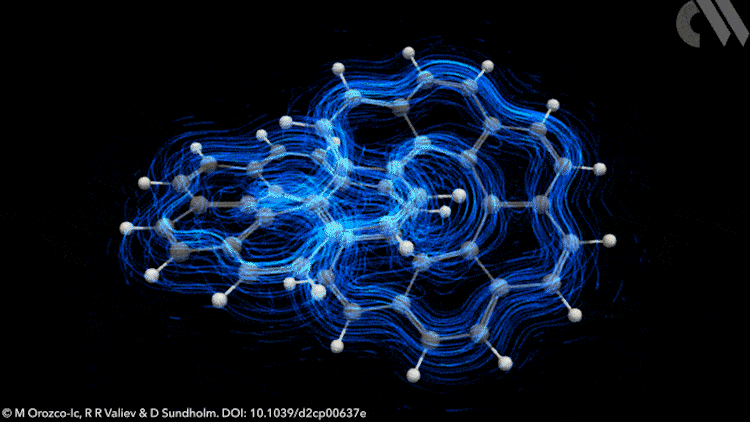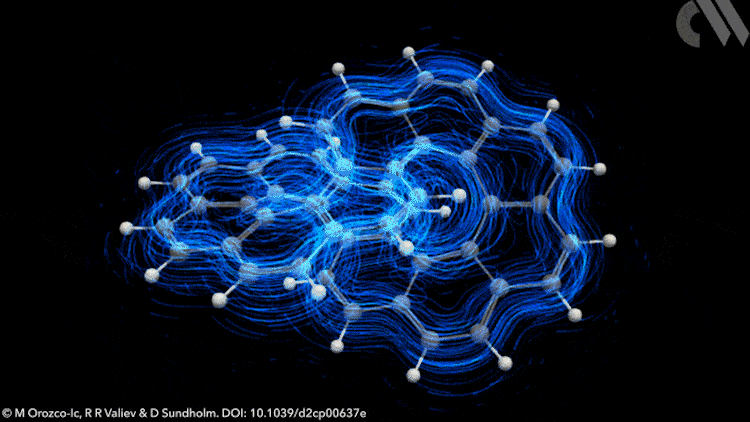
However, applying Möbius rules to the π-stacked systems seen in polyarenes like infinitene is not straightforward. Now, new calculations show that infinitene produces two separate current-density pathways of different sizes that flow along the edges of the molecule in response to an external magnetic field. These current loops are diatropic, meaning their induced magnetic fields oppose the applied field as they pass through their respective current loops.
Crucially, these current-density pathways do not intersect. Rather, they create a shielding cone over the whole structure, suggesting infinitene is, in fact, aromatic. The researchers also demonstrate the antiaromaticity of the infinitene dication using a similar approach, a result that is equally unexpected.
The contrast of these findings to the Möbius rules highlights that simply counting the number of π electrons in new polyarenes with similarly intriguing topologies should be treated with caution.
References
1 M Orozco-Ic, R R Valiev and D Sundholm, Phys. Chem. Chem. Phys., 2022, 24, 6404 (DOI: 10.1039/d2cp00637e)
2 M Krzeszewski, H Ito and K Itami, J. Am. Chem. Soc., 2022, 144, 862 (DOI: 10.1021/jacs.1c10807)








No comments yet