US researchers have demonstrated that an iridium pincer complex can stoichiometrically strip carbon monoxide from a wide range of ethers to give two hydrocarbyl products. The unique chain-rupturing transformation involves breaking an extraordinary number of bonds.
Alexander Miller and his colleagues at University of North Carolina at Chapel Hill were originally interested in adding carbon monoxide to molecules, as part of a project sponsored by Eastman Chemical, when they made a serendipitous discovery. ‘Changho Yoo, a postdoc in my group, wanted to prepare an iridium(I) precursor for those types of reaction. His first reaction generated a mixture of products and it didn’t look very promising. I think a lot of people would have walked away at that point but Changho felt like there was something interesting there,’ says Miller.
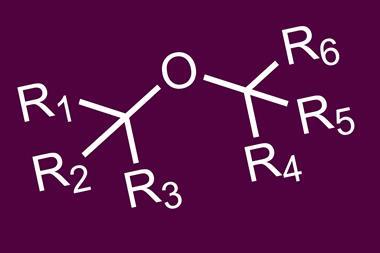
Rather than adding carbon monoxide, Miller’s team uncovered a process that cleaves ethers into carbon monoxide and hydrocarbyl products. Ethers are present in a wide range of organic compounds, as well as functional polymers and biomass, and forming two saturated hydrocarbyl products from ethers is unprecedented. The ethers are selectively dissected by cleaving two C–H bonds, one C–C bond and one C–O bond. Two new C–H bonds are formed, resulting in the hydrocarbyl products and carbon monoxide, which is directly coordinated to the resulting iridium species. Mechanistic studies indicate that the hemilability of the amine ligand is fundamental in controlling the reaction pathway. The decarbonylation reaction can be intramolecular and generates a linear (poly)ether product from a crown ether, but intermolecular decarbonylation is also possible.
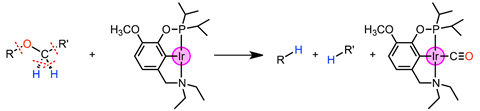
Although carbon monoxide releasing reactions are widely known for organic carbonyl compounds, decarbonylations of ethers are rarely reported. The findings could therefore provide opportunities to activate a range of useful ether starting materials. And although yields are currently low, the process can coax even simple and highly stable ethers such as diethyl ether and tetrahydrofuran to react.
Robin Perutz, an organometallic chemistry expert at the University of York in the UK, says the reactions are very unusual. Since ethers are ‘one of the more recalcitrant functional groups, these reactions have potential for application.’ ‘Organometallic chemistry continues to have potential for astonish. My initial surprise was tempered by the demonstration that the reaction is initiated by C–H bond activation followed by hydrogen migration to form a carbene complex, an analogous sequence to that reported in several other reactions of ethers at iridium. The remarkable feature is that this sequence is followed selectively through to products at modest temperatures.’
The team is now working to develop the system to have catalytic turnover. ‘There are certainly opportunities using photochemistry, or high temperatures or flow conditions,’ says Miller. ‘I think it’s a matter of finding the right system that can do the reaction of interest, but then has the appropriate stability to either be heated to high temperatures or to undergo a photochemical reaction.’
‘One of the most exciting directions would be degradation of polymer and biomass,’ adds Yoo. ‘Ether functional groups are very common in those molecules and it is generally considered as a stable functional group. However, with this decarbonylation, they aren’t anymore!’








No comments yet