Domestic sponge fortuitously found to soak up bisphenol A

Scientists have discovered that a cheap and common sponge they use to clean surfaces and equipment in their lab has a very high capacity to absorb bisphenol A (BPA).
‘It was an accidental discovery,’ says Wei Qiu, from the University of Massachusetts, US, one of the researchers involved. ‘There was a big tank of waste BPA solution and while we were testing some other absorbent materials we accidentally dropped a sponge into the solution. We were curious and when we tested the waste solution we found a significant drop in BPA concentration and the only thing that could account for that drop was the sponge.’
BPA is a building block for polycarbonate plastics and epoxy resins and, despite currently being phased out of industrial processes, has become a significant pollutant in industrial wastewaters. Environmental and health concerns associated with BPA have drawn researchers to find methods to reduce its presence.
The polyurethane sponge has fibrils that swell when they absorb liquid. Carbonyl and polyol groups on the fibrils hydrogen bond with guest BPA molecules diffused into the foam.
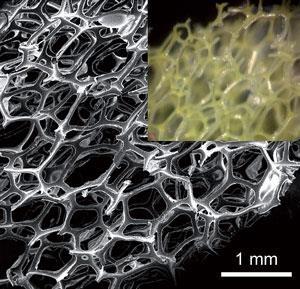
Lead scientist Jie Han explains how they recycle the foam: ‘We use an alkaline solution to deionise the hydrogen bonded BPA, which can then be washed off the material and recovered by neutralising the resultant alkaline solution. Compared with other sorbents like activated carbon or polymer resins this process is really easy.’ The sponge could be used up to 25 times with no sign of deterioration.
It is possible to make the foam in a variety of sizes, geometries and densities making it suitable for various sorption processes. ‘One particularly good thing about this material is its abundance and low cost,’ says Qiu, ‘combining its high flow rate and high sorption rate produces a very high treatment capacity, which is exciting for real application.’
Environmental materials scientist Saravanan Pichiah from the University of Malaya, Malaysia, comments that this kind of work ‘is an awakening idea for the implication of such commercial grade polymeric materials for environmental cleanup.’
References
This article is free to access until 26 May 2015. Download it here:
J Han et al, J. Mater. Chem. A, 2015, 3, 8870 (DOI: 10.1039/c5ta00868a)












No comments yet