Grinding solid materials in a ball mill speeds up sublimation and can be used to separate chiral molecules in a simple way. The finding by scientists in Germany who developed a way to monitor the process in real time using NMR spectroscopy, could open up new possibilities in pharmaceutical research and medicinal chemistry.
‘We introduced a new tool for controlling the way solids turn into gases,’ says Carsten Bolm from RWTH Aachen, who led the project. ‘This has implications for product purification and enantiomer enrichments of chiral compounds, perhaps even impacting questions related to the origin of enantioselectivities in prebiotic life.’
Bolm notes that the approach is based on previous work carried out by US researchers in 2007, who found that a-(trifluoromethyl)lactic acid (TLA) showed an unusually high evaporative sublimation rate. Molecular recognition then led to optical self-purification, producing enantiomerically enriched samples.

‘Although this approach has been known for years, we feel that it hasn’t received enough attention,’ says Bolm. ‘We now looked into more details of this process and were able to accelerate and control the sublimation by fine-tuning the milling conditions.’
Bolm’s team are convinced that the TLA experiments show that solid–solid interactions are the main factor influencing the sublimation properties of materials. ‘While in this case two enantiomers of the same compound recognised each other in the solid state, such self-finding processes can also involve two different chiral molecules,’ clarifies Bolm. ‘Thus, in subsequent studies, we manipulated the sublimation speed of D-(+)-camphor through its interaction with another chiral solid, which then affected the transition of camphor from the solid to the gas phase.’
Team member Francesco Puccetti explains that the experimental setup included a ball-milling machine, which allowed controlled grinding and mixing of the solids, attached to a benchtop NMR instrument.
‘A very particular vessel with gas in- and outlets, designed by Krunoslav Užarević and colleagues at the Ruđer Bošković Research Institute in Croatia, enabled us to establish a constant flow of a chemically inert gas – argon or nitrogen – while grinding the solids,’ he says. During sublimation, the gas was enriched with the compound of interest and after leaving the vessel, it was trapped in dimethyl sulfoxide for real-time analysis.
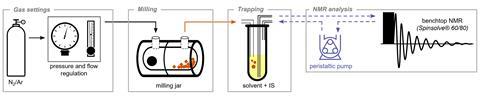
Tomislav Friščić, an expert in mechanochemistry at the University of Birmingham, UK, who wasn’t involved in the study, is impressed by the results. ‘What’s truly outstanding is that the researchers were not only able to ingeniously monitor and measure the vaporisation of the solids that were being ground but could also establish that this vaporisation leads to spontaneous separation of molecules which are mirror images of each other,’ he says. ‘This could become an entirely new way – also clean and solvent-free, so quite safe – to separate biologically important molecules.’
But the work is also relevant from a fundamental point of view, adds Friščić. ‘For me as a materials scientist and physical chemist, this is a really interesting new tool to study what happens when we mechanically grind and crush materials.’
Pedro Cintas at the University of Extremadura in Spain says that this is the first reliable NMR monitoring system for mechanochemically enhanced sublimations that can ‘disentangle the fate of analytes through the air–solid interface and screen the nature of favourable intermolecular interactions’ during the process.
‘There’s a couple of really interesting aspects to this setup, which makes me want to have one in my lab,’ comments Friščić. He believes that the main challenge was to get the gas to flow through the reaction vessel. ‘That’s a big problem because milling jars move very quickly and it’s difficult to build connections to such rapidly moving instruments.’
The researchers are now planning to improve the system further by automating the gas-flow regulation and developing ways to control the milling jar and gas temperatures.
References
Puccetti et al, Cell, 2023, DOI: 10.1016/j.chempr.2023.02.022









No comments yet