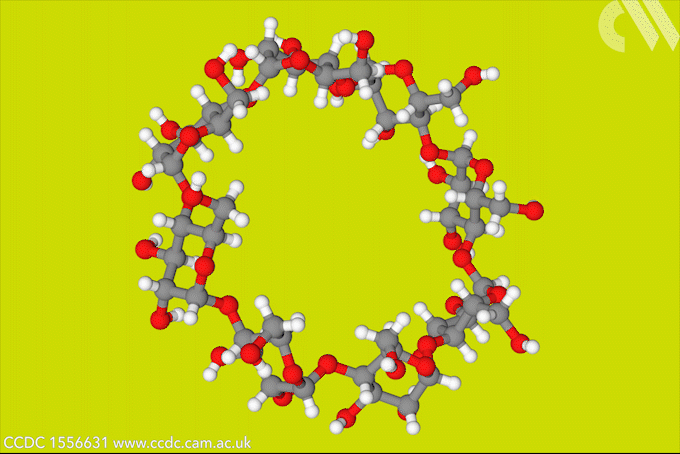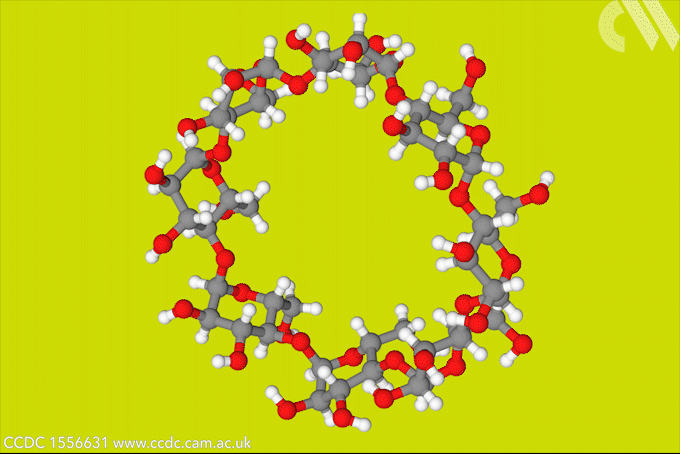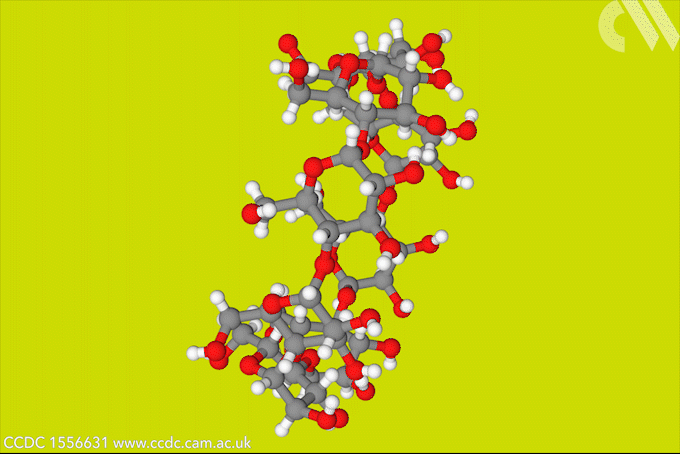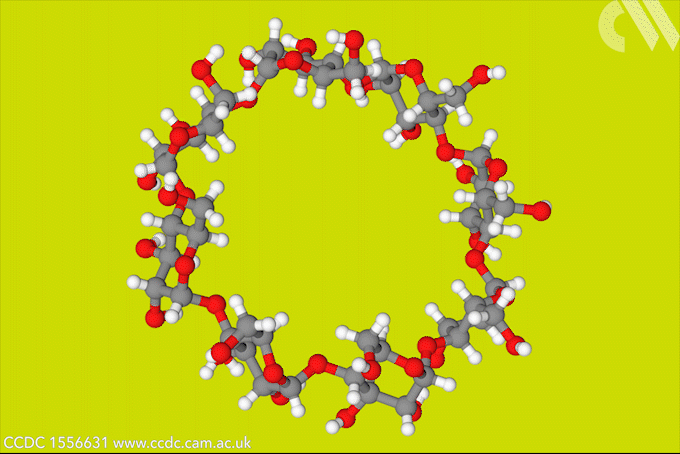
A light-activated molecular switch could drive down the cost of a useful, but difficult to make γ-cyclodextrin.
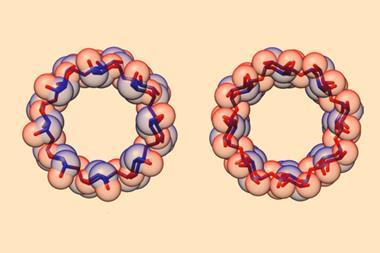
Cyclodextrins are ring-shaped structures typically made from six to eight glucose units. Their hydrophilic exterior and hydrophobic cavity makes them important host molecules used in supramolecular chemistry, with applications ranging from food chemistry to drug delivery.
γ-cyclodextrin, made from eight glucose units, can fully encapsulate bulky compounds like vitamins. But its high water solubility makes it tricky to synthesise. The energy-consumptive steam distillation process required to isolate γ-cyclodextrin means that it is several times more expensive than smaller cyclodextrins.
Now, a team of researchers from Denmark and the US has developed a light-controlled synthesis that could make accessing γ-cyclodextrin much easier. The strategy relies on a hydrazone template, which switches between E- and Z-isomers in response to light. The team took advantage of the fact that the Z-isomer binds to γ-cyclodextrin to stabilise the material during an enzymatic synthesis, bringing about a six-fold increase in yield. Because the E-isomer has a much lower binding affinity, the team could then use light to release the cyclodextrin product.
The researchers hope that their simplified synthesis will ‘open the way for designing new γ-cyclodextrin adaptive self-assembled materials’.
References
S Yang et al, Chem, 2021, DOI: 10.1016/j.chempr.2021.05.013
















No comments yet