At the hands of Ernst Beckmann, two major chemical developments came from isolating some very simple compounds.
At the hands of Ernst Beckmann, two major chemical developments came from isolating some very simple compounds, says Colin Russell
Most people like to have things named after them. In the UK hundreds of streets and public buildings testify to royal, political and military figures once counted deserving enough to lend their names. Scientists don’t fare quite so well, though the biologists can point to Robinson’s skua, Cuvier’s whale and roses named after almost anyone, while the physicists have laws attributed to Newton, Faraday, and Ohm. And there’s always Halley’s Comet. Chemists, however, are even less fortunate and many giants are forgotten just because no one thought to commemorate them by a law or a piece of apparatus. Lavoisier, Berzelius and Frankland are just three examples.
But one chemist had the good fortune to give his name to both an important piece of apparatus and a significant reaction. He was born 150 years ago and is one of several chemists called Beckmann (or something similar). This one was Ernst Otto Beckmann, born on 4 July 1853 in the small German town of Solingen. From him we derive the Beckmann thermometer and the Beckmann rearrangement. He also did much other excellent work that bears his name as author. He was the son of Johannes Friedrich Wilhelm Beckmann who owned a small dyestuffs factory. Nearly 30 years before his son was born Johannes had discovered Paris Green, a brilliant pigment and insecticide, being a double compound of copper arsenate and acetate. Young Ernst grew up in a place where no one minded getting his hands dirty in experiments, and where chemistry exerted a strong appeal.
His schooldays over, he spent five years in varied laboratory activity, including one year with the great analytical chemist Fresenius in Wiesbaden.
Student years
Rather later than usual Beckmann entered university, at the age of 22, spending the next three years as a student of the famous chemical pioneer Hermann Kolbe in Leipzig. Such was his grasp of chemical techniques that he proceeded almost at once to research. By this time (1875) Kolbe’s health was deteriorating and his assistant, Ernst von Meyer, was largely responsible for the day to day supervision of research students. Under his guidance Beckmann studied the oxidation of dialkyl sulphides to sulphones (for example with permanganate) and gained his PhD in 1878.
The legacy of Kolbe included a profound belief in the value of organic research, coupled with a certain agnosticism towards the new structural theories that had been in vogue for the past 10 years. Kolbe played a minor role in Beckmann’s supervision, and Beckmann was one of only three of Kolbe’s students to rise eventually to the rank of full professor, yet he always entertained a great respect for his mentor and was clearly influenced by his values.
After graduating at Leipzig, Beckmann became assistant to the toxicologist Robert Otto at Brunswick. Here he did some work on barium halides and aluminates and developed an interest and skill in pharmaceutical chemistry, though it seems that his work was less than fulfilling. After five years at Brunswick an opportunity arose for him to return to Leipzig. What one biographer called his Wanderjahre, or ’years of wandering’, were over.
Warmly welcomed back by Kolbe, Beckmann began work on menthone, which he made by gentle oxidation of menthol. From this and similar ketones he obtained oximes by treating with hydroxylamine, and concluded they were isonitroso compounds, containing the group >C=N-OH. These quite ordinary compounds were then suddenly invested with a new significance when Beckmann, in 1886, discovered the action of acids upon them. The product was a substituted amide. To form this, Beckmann assumed an intermediate in which one alkyl group changed places with the hydroxyl (Scheme 1).
Scheme 1. Beckmann assumed that an intermediate was formed during the reaction of isonitroso compounds [oximes] with acids. This overall reaction became known as the Beckmann rearrangement
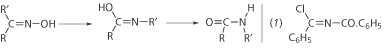
His hypothetical intermediate was justified by the isolation of what he called an iminochloride (1) from the monoxime of benzil, using phosphorus pentachloride with rigid exclusion of moisture. All kinds of acids would have this effect on oximes, but it was best accomplished with phosphorus pentachloride in dry ether. He found that even acetic anhydride or acetyl chloride might not acetylate all the oxime but might also yield small amounts of substituted amide.
Rearrangement reaction
The discovery of the Beckmann rearrangement reaction, as we now know it, came at a crucial moment in the history of organic chemistry. Until then very few reactions were recognised in which a group or atom changed places with another within the same molecule. Put differently, we could say that in nearly all known reactions the basic framework of the molecule, the carbon chain, remained intact. Thousands upon thousands of new organic compounds had been synthesised on the assumption that this was true. A mere handful of possible exceptions remained in need of explanation. They included Liebig’s benzil-benzilic acid transformation (1838), Fittig’s pinacol-pinacolone interconversion (1860), and the famous degradation of amides to amines found by Hofmann (1881). Now here was something that seemed to be another exception, and quite different from the normal experience in organic chemistry. Though he could not know it, the reaction discovered by Beckmann was to tantalise generations of organic chemists, and only recently has its mechanism become clear.
It was perhaps fortunate that, at about this time, Kolbe was succeeded by Johannes Wislicenus, the man who recognised and named the phenomenon of ’geometric isomerism’ as seen in maleic and fumaric acids. By now the numbers of oximes turning up in reactions was causing increasing concern. Benzil, for instance, seemed to produce three dioximes. A current explanation was that, for some unexplained reason, there was restricted rotation about the central single bond and the molecule got stuck in one of three configurations. But shortly after this (1889), Beckmann discovered that the simple molecule benzaldehyde gave rise to a pair of oximes, and that could not be due to that kind of restricted rotation. It seemed to Hantzsch that these might be geometric isomers, like those with carbon-carbon double bonds, and the benzil dioximes could be similarly explained (Fig 1).
Fig 1. The geometrical isomerism of the oximes of benzaldehyde and the dioximes of benzil

Thus, on the basis of Beckmann’s work, Hantzsch proposed that the valencies of nitrogen were not confined to one plane but were like those of carbon and thus capable of giving rise to a new kind of geometrical isomerism.
Complicated history
The subsequent history of the Beckmann rearrangement was truly complicated. On the basis of his assumption, Hantzsch proposed that the groups exchanging places were those nearest each other in space - in other words a cis migration. This was disproved in several ways, most notably by Meisenheimer in 1921 who found that ozonolysis of triphenyliso-oxazole led to a benzoylated benzil monoxime which must therefore be of trans configuration (Fig 2a). The oxime derived from this product was then known as the ?-monoxime, and on Beckmann rearrangement it yielded a product that clearly implied a trans, not cis, migration (Fig 2b).
Fig 2. In 1921, Meisenheimer discovered that the Beckmann rearrangement involves a trans migration
(a)

(b)
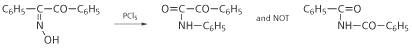
Subsequently this was confirmed by evidence from many diverse sources (ring closures, dipole moments, steric hindrance etc). The ionic character of the reaction had been postulated by Beckmann, and is now widely accepted, as is its intramolecular nature. Yet it has not simply served to puzzle theoretical organic chemists for over a century, but has been useful in a multitude of syntheses, particularly of amines and their derivatives.
Just as the 1880s were a time when awkward new problems (like molecular rearrangements) reared their head, so it was a period with a great deal of unfinished business. Most notably this concerned molecular weights (masses). The figures for gases and volatile liquids had been derived from vapour density data for some time, and few chemists after 1860 doubted these. However the question of molecular weights for solids had not yet been solved and no one could be sure that one of Beckmann’s oximes might not have been, in today’s terms, a huge macromolecule. It was pure assumption that simply because a ketone might be known to occur in monomeric form the same was also true of its oxime. To be sure it was the simplest view, and not a bad working hypothesis, but that did not guarantee its truth. So molecular formulae like those above were essentially acts of faith rather than statements of fact.
To those of us who instinctively turn today to the nearest mass spectrometer for information on molecular mass it is hard to imagine just how difficult organic chemistry was for people like Beckmann. Yet if the problem of molecular weights of dissolved solids had not been solved in the 19th century the data would not have been available to make modern techniques possible, let alone credible.
Fortunately Beckmann recognised the problem. While still researching on oximes Beckmann had included studies of their physical properties, and these together with the analytical training from Fresenius, were now to be reinforced by F. W. Ostwald who came to Leipzig as professor of physical chemistry in 1887. The laws relating colligative properties with molecular masses offered hope that the new physical chemistry might be useful even to organic chemists (who generally regarded it with suspicion). These colligative properties depend on the number, not the nature, of the particles dissolved. They could make it possible to determine the number of molecules present in solution, and given their total weight one could calculate their molecular mass.
New thermometer
A link between depression of freezing-point and mass had been recognised by Richard Watson (1771) and by Sir Charles Blagden (1788), and it was exactly a century after the announcement of Blagden’s Law that Beckmann began detailed studies of this relationship. Realising that contemporary thermometers were hopelessly inaccurate for the task, he designed a new one. The Beckmann thermometer had a mercury reservoir at the top so that the amount of mercury could be varied. It did not measure absolute temperatures, but rather temperature differences, which is what was needed for precise measurement of freezing-point depressions. He was able with this device to measure to within 0.001?C. He became the founder of the cryoscopic method for molecular mass determinations - the Beckmann method. Soon afterwards he tried the application of Raoult’s Law (1871) to determine vapour pressure changes, but quickly realised that boiling-point elevations were far easier to measure. So, again using ’his’ thermometer, he devised a further technique, molecular weight determinations by ebulliometry or boiling-point elevation, though it was never as useful as the cryoscopic method.
Beckmann’s final publication from this Leipzig period reported the formation of a dark blue solution when sodium was added to benzophenone in dry ether. He had discovered the metallic ketyls.
Truly his activity between 1885 and 1890 led to permanent changes in chemical theory and practice. It marked the end of an era for him, and in 1891 he was appointed extraordinary professor at Giessen where he continued his oxime research. It was to be a brief interlude before his appointment as full professor at Erlangen. Here his long-standing interest in terpene chemistry led to advances in the camphor series and the production of menthol by reduction of menthone. In 1897 he returned for the third time to Leipzig, now as professor of applied chemistry. All his previous themes were continued, also incursions into the new field of spectroscopy, recognition of sulphur tetrachloride and the resolution of camphoric acid. His new applied chemistry laboratory was so splendidly equipped that he wrote a book about it.
Final research
For 15 years Beckmann remained at Leipzig until, in 1912, he accepted a call to become director of the new Kaiser-Wilhelm Institute in Berlin. By now he had emerged as a strong critic of the long-established German practice of research by large numbers of undergraduates. In Britain and France he saw how professionally trained chemists, Ramsay and Curie, for example, were so much more effective. This is a powerful corrective to the common view that, before 1914, Germany’s chemical supremacy was accepted by all. International competition was intensifying, the US being seen as the most dangerous of Germany’s rivals. So Beckmann argued ’In the peaceful competition of nations Germany can conquer only in the sign of scientific and industrial progress’.
The Prussian government knew he was their man, and before long he was designing yet another magnificent laboratory for Berlin, helped by R. Willst?tter. When war came in 1914 most of his students and assistants left for military service. Some research was dictated by wartime needs, but for the most part Beckmann quietly pursued his own investigations, chiefly in ebulliometry. His final shots in research included inventing a new sodium press and studying organic products from lupins. Honoured by governments of Saxony and Prussia, magnificently bearded and looking like ’an old Thuringian sovereign’, in April 1923 he showed symptoms of pernicious anaemia and died three months later.
Source: Chemistry in Britain
Acknowledgements
Colin Russell is emeritus professor in the department of history of science at the Open University, and affiliated research scholar at the department of history and philosophy of science, University of Cambridge.
Further Reading
- G. Lockemann, Ber., 1928, 61, 87A.
- J. A. Johnson, The Kaiser’s chemists. Chapel Hill: Univ. N. Carolina Press, 1990.






No comments yet