A new technique overcomes a major problem with the production of zeolite nanosheets
US scientists have overcome a significant hurdle in the production of zeolite nanosheets, which should make these versatile materials simpler to synthesise. Zeolites nanosheets could potentially be used for a wide range of applications such as sieves that can separate molecules based on their size and catalytic membranes.
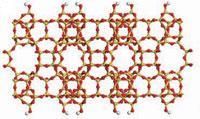
Presently, synthesis of zeolite nanosheets is hampered by one crucial step. The exfoliation stage which is required to produce discrete nanosheets is plagued by aggregation of the sheets and structural deformation of the zeolite framework, including the formation of pinholes which are larger than the zeolite’s pores.
Now, these obstacles have been overcome by a new procedure developed by Michael Tsapatsis and colleagues at the University of Minnesota. The team manufactured the layered zeolites silicalite and swollen ITQ-1 and then melt blended them with polystyrene to create nanocomposites containing nanosheets with two different structures - MFI and MWW, respectively.
Attempts to remove the polystyrene matrix from the nanocomposites by calcination and other thermal treatments were unsuccessful due to the formation of curled particles, which are unsuitable for subsequent coating. The researchers got around this problem by sonicating the nanocomposites in toluene and centrifuging to remove larger particles to create flake-like, crystalline nanosheets. They displayed uniform thicknesses of 2.6 and 3.4nm for MMW and MFI, respectively.
The nanosheets were used to coat an anodised alumina membrane and a rough alpha-alumina support to produce zeolite membranes. Subsequent calcination gave smooth, well packed overlapping layers.
Ryong Ryoo, from the Korean Advanced Institute of Science and Technology (KAIST), who prepared the first MFI zeolite nanosheets in 2009 says: ’The authors seem to have much less of a problem with pinhole generation. This is because the zeolite nanosheet of single unit cell thickness is somewhat flexible and can be overlappingly coated onto a porous support even in the case of surface curvature.’
The coated structures displayed no selectivity as molecular sieves for o-/p-xylene until they had undergone hydrothermal treatment to reduce the space between the nanosheets. After this treatment the nanosheets were highly effective at separating xylene isomers. Good performance was also observed for helium-hydrogen and helium-nitrogen mixtures.
Tsapatsis says that the technique should be applicable to any zeolite or other molecular sieve structure that can be prepared as a layered material. Although he adds: ’I do not know if all these other available materials will exhibit the same degree of framework preservation as the two we used up to now.’
Steve Down
References
K Varoon et al, Science, 2011, 334, 72 (DOI: 10.1126/science.1208891)




No comments yet