Researchers in the US have performed an electric field-driven chemical reaction in bulk solution. It is the first study to quantify chemical rate enhancement by an external electric field and paves the way for chemical reactions that no longer require chemical reagents or harsh conditions to drive them.
Electric field-driven chemical reactions are a promising sustainable solution to current catalytic processes. They could omit the need for chemical reagents or catalysts, and the production of waste products, whilst achieving the same reaction rate and having more control. The idea of using an external electric field to catalyse a chemical reaction has been theorised before, but actually applying this experimentally has only occurred recently. Now, Latha Venkataraman and her research group from Columbia University have used the scanning tunnelling microscope-based break-junction technique (STM-BJ) to induce an external electric field to drive the homolytic cleavage of the O–O bond of a benzoyl peroxide to form a benzoic acid in a bulk solution.1 ‘From a green chemistry and sustainable chemistry point of view this work is very important,’ comments Deqing Zhang from the Institute of Chemistry Chinese Academy of Sciences, an organic materials expert who also uses the STM-BJ technique.
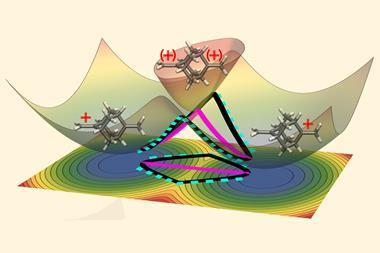
You might expect that polarising a chemical bond with an electric field would cause the bonding electrons to move to just one atom involved the bond. However, using DFT calculations, Venkataraman’s team found that introducing the electric field to the peroxide bond caused a population transfer from the bonding to the anti-bonding orbital, weakening the bond and encouraging it to break into two thermodynamically stable radicals. Characterisation, using high performance liquid chromatography and UV–vis analysis, confirmed the products from the electric field-driven homolytic cleavage and quantified the reaction kinetics.
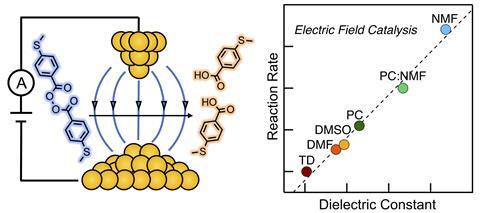
After performing the chemical reaction in solvents with differing polarities, the group discovered that the chemoselectivity of the bond cleavage was controlled by the solvent environment. Polar solvents yielded the desired product, a benzoic acid, and non-polar solvents yielded a biphenyl. Surprisingly, as the dielectric constant of the solvent increased, the rate at which the reaction proceeded in an electric field also increased. ‘This was completely counterintuitive, and I have to honestly say that we don’t completely understand the result but I’m an experimentalist at heart and so we do the measurement, we report what we measure and then we try and explain it,’ Venkataraman tells Chemistry World.
The STM-BJ technique has also recently been used by a different research group in China to catalyse a C–C bond cleavage reaction using an electric field. The linear oligophenylene products from that reaction were covalent adducts on the gold surface of the STM tip.2 The work from Venkataraman’s group ‘is one of the first examples where the substrates are not permanently immobilised on the gold surface,’ says Rose Kennedy, who studies how field effects influence reactions at the University of Rochester in the US. Venkataraman’s work ‘provides a proof-of-principle for applications on bulk solutions, suggesting future opportunities for scalability of field-modified reactions,’ Kennedy adds.
‘If you had asked me when we had written our proposal for our [work], I would’ve said that these are two things that we won’t observe, yet here we are,’ says Venkataraman. With these surprising results in hand, the Venkataraman group plan to continue its work on electric field driven strategies by investigating other chemical reactions.
References
These articles are open access
1 B Zhang et al, Chem. Sci., 2023, DOI: 10.1039/d2sc06411a
2 J Lin et al, Nat. Commun., 2023, 14, 293 (DOI: 10.1038/s41467-022-35686-4)










No comments yet