Reaction efficiency could be improved by exploiting vibrational excitation
A chemical reaction has been sped up simply by using pulses of infrared (IR) light to excite molecular vibrations. The approach could replace the need for heat in some thermally driven reactions, making them more efficient, as well as offering a new cost-effective photolithography technique.
Many chemical reactions require heat and are, therefore, manipulated by changing temperatures. However, such changes influence all molecules and all reaction processes. In a complex synthesis, this makes it difficult to control competing reaction pathways that produce unwanted products thereby decreasing yields of the desired product.
The beauty of the result is the tantalising hint of the possibility of exploiting vibrational excitation to control reactions
Fleming Crim, University of Wisconsin–Madison
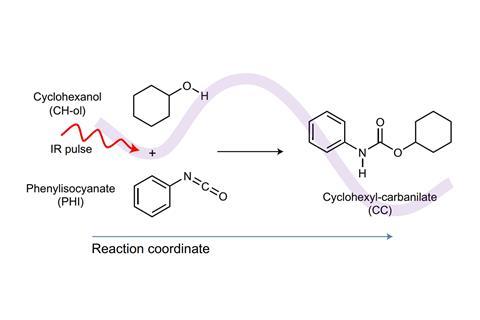
Now, Karsten Heyne at the Free University of Berlin, Germany, and his colleagues have shown that ultrafast pulses of IR light can steer a reaction that’s usually thermally driven and accelerate it. As IR can target and excite specific molecular vibrations that are closely associated with a reaction pathway without influencing other molecules normally affected by heat, the approach made one reaction up to 24% faster.
’We show for the first time that IR excitation can accelerate a ground state synthesis reaction in solution between two molecules,’ explains Heyne. ‘Since we can influence specific reaction coordinates, our method allows to increase or decrease wanted or unwanted reaction pathways. This should open up new strategies to optimise all thermally driven chemical reactions, such as those for antibiotics, and thus reduce the costs.’
Theoretical predictions suggested that IR excitation of specific bond vibrations could accelerate thermally driven reactions by stretching bonds to provide new conformations that help the reaction along. But until now researchers had been unable to demonstrate it. That’s because they focused on breaking bonds with IR to trigger a reaction, but since the lifetime of an IR excited vibration is incredibly short – around one quadrillionth of a second – it was too hard to stop other processes from diverting the excitation energy needed for the trigger.
To overcome this, Heyne’s team concentrated on a different type of reaction – namely two bimolecular reactions in urethane and polyurethane formation. Since these occur by themselves at room temperature, they just needed IR pulses to excite the bond vibrations to initiate the reactions. This type of reaction also meant the team could achieve the signal-to-noise ratio necessary to identify the IR-induced product based on the absorption of IR pulses. Following this, the team even demonstrated the technique’s potential as a photolithography tool by using an IR laser to draw polyurethane squares.
‘I am struck that they can observe the acceleration in the presence of significant background [noise],’ comments Fleming Crim, who investigates reaction dynamics at the University of Wisconsin–Madison, US. ‘They have overcome a major obstacle in making that measurement. The beauty of the result is the tantalising hint it provides of the possibility of exploiting vibrational excitation to control reactions in solution in practical systems,’ he says.
References
T Stensitzki et al, Nat. Chem., 2017, DOI: 10.1038/nchem.2909












No comments yet