Disagreements over the definition of a chemical step underlie much broader questions
What makes a chemical step? You might think the answer is intuitively obvious: take some stuff, turn it into some other stuff, and there’s your step. But a recent discussion on the Open flask blog, owned by Phil Baran’s group at the Scripps Research Institute in California, US, shows that it’s not so simple. Not by a long shot.
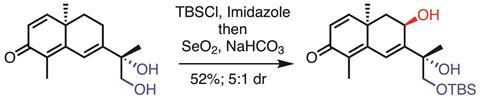
The debate centered on the group’s total synthesis of (–)-thapsigargin, a complex molecule bristling with stereocentres that was first discovered in a plant with the unlikely name of Deadly carrot. Previous syntheses have clocked in at 36 to 42 steps, but in December Baran’s team unveiled a remarkable 11-step synthesis.1
Their submitted paper originally billboarded this step-count in the title, but in the published version it was buried towards the end of the paper. That was in response to feedback they had received during peer review, which boiled down to these critiques: step count is a poor metric to judge a synthesis, and Baran repeatedly under-counts his steps anyway. Ouch.
This might seem like no more than an academic spat. But if you pull on this thread, the dispute over step count leads to important questions about nothing less than the future of total synthesis.
Watch your step
There’s one particular part of the team’s thapsigargin synthesis, involving a diol, which illustrates the step count conundrum. First, they protected one of the diol’s –OH groups; then they added more reagents to the crude reaction mixture to install a new –OH group elsewhere in the molecule.
Critics say this should count as two steps, because it involves two different reactions on two different functional groups. Baran insists that it’s one step, because a step only comes to an end when there is some kind of work-up or purification. He also points out that there are plenty of reactions that everyone agrees are a single step, yet involve multiple transformations around different parts of a molecule – just think about the amazing cascade reactions that can zip up natural product ring systems.
A straw poll about this on the Baran lab’s Twitter feed garnered almost 500 responses, with 73% agreeing with his point of view. But that is a long way from consensus. One alternative view is that bundling reactions together like this might be better described as ‘one pot’ rather than ‘one step’. Another is that failing to purify and characterise all stable intermediates in the sequence amounts to corner-cutting. A third, frankly jaw-dropping, suggestion is that it unfairly belittles previous synthetic routes that were not focused on being as short as possible.
Step count is not a perfect metric, to be sure. There is artistry in total synthesis, and sometimes a meandering journey is more rewarding than the shortest route possible. But synthetic chemistry is ultimately a practical science, and we need some way of telling if one route is better than another – not for bragging rights, but for sheer practicability.
Baran’s lab explicitly aims to make grams of products using short, efficient routes that can be scaled up. Step count provides a reasonable rule of thumb for a bench chemist following their recipes, because it gives a sense of how much time-consuming purification they’ll have to do. At a time when some claim the pharmaceutical industry is no longer interested in complex natural products because they are too laborious to synthesise, Baran is aiming to prove them wrong.
The next step
This feeds into a much broader debate about what makes an ‘ideal’ synthesis. Step economy might be a useful target, but it is far from being the only way of measuring ideality. A process chemist might be more interested in the total number of operations in the synthesis. An ‘atom economy’ approach aims to ensure that each reagent contributes as many atoms as possible to the final product, to minimise waste. And Baran take pains to avoid using protecting groups and unnecessary redox reactions to maximize the proportion of reactions in a synthesis that are constructing the product’s skeleton.
Meanwhile, as the principles of green chemistry become increasingly integrated with everyday synthesis, they bring with them a portfolio of other metrics, from environmental impact factor to process mass intensity. There is a growing need for the community to settle on some common standards. Blogs like Open flask, which was set up to provide as much transparency as possible about the group’s work, can only go so far in stimulating the discussion. This is a debate that should involve the wider community of synthetic chemists, journals and learned societies.
Forty years ago, grinding out a few milligrams of a fiendishly complicated natural product for characterisation was, given the chemical toolkit available to practitioners of the time, a tremendous achievement. Today, though, more and more chemists are aiming to reach those pinnacles in ways that are as quick, cheap, efficient and scalable as possible. This shift in emphasis raises questions about the key goals of total synthesis in the 21st century. Defining that vision in measurable ways may be the most important step of all.
References
1 H Chu et al, ACS Cent. Sci., 2016 DOI: 10.1021/acscentsci.6b00313











No comments yet