Gold cluster bends between two isomers
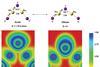
Unusual structural isomerism uncovered with photoelectron spectroscopy and theoretical calculations

Unusual structural isomerism uncovered with photoelectron spectroscopy and theoretical calculations