Scientists make longest heparin-related oligosaccharide to date
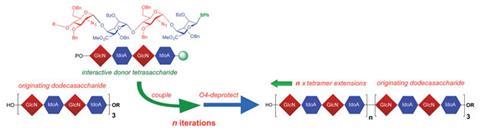
The traditional synthetic route to long chain oligosaccharides is drawn out and complicated; they require numerous steps to produce the precursor tetrasaccharides, then further steps to connect these precursors into long chains. Normally they’re made by an iterative two block process, generating chains up to 12 units long. Now, a four block approach, developed by John Gardiner and colleagues at the University of Manchester, significantly reduces the number of steps enabling longer sugar lengths from 16 up to 40 units. ‘Our solution-phase method requires only simple lab techniques to make the oligosaccharides,’ explains Gardiner, opening up further research possibilities to explore their biological significance and therapeutic potential.
‘Glycosaminoglycans, typified by heparin and heparin sulfate, have functions in a wide range of biological processes and are extremely difficult to obtain from nature in homogenous form,’ says Rob Field, who leads a biological chemistry group at the John Innes Centre in Norwich, UK. Gardiner’s technique means they can now be made synthetically in large amounts at high purity so researchers can better explore their properties and find potential applications.
Jacquelyn Gervay-Hague, a synthetic chemistry expert at the University of California, Davis, US, says ‘this is an impressive example of how building on previous work – in this case establishing the viable gram scale syntheses of the building blocks – can lead to extraordinary achievement.’ And synthetic oligosaccharide chemist Carmen Galan, from the University of Bristol, UK, says ‘the synthetic methods developed in the context of this work have the potential to be applied to other relevant carbohydrates, which is very exciting,’
References
This article is open access. Download it here:











No comments yet