One-time heavy rocker Carolyn Bertozzi has made a name for herself with hit papers and sound science. Sarah Houlton charts her path to success
US chemist Carolyn Bertozzi reckons her path into science was almost subconscious. Her dad was a physics professor at the Massachusetts Institute of Technology but when she started at nearby Harvard University she was tempted to major in music. ‘That was unpopular with my parents – a cousin had pursued a career in music and ended up working in a bank to pay the rent,’ she says. ‘I was a little timid about defying them.’ Instead, she chose the pre-med track that included classes in maths and the sciences, and declared herself a biology major at the end of her first year at college. ‘I didn’t really enjoy the general chemistry class in my freshman year,’ she claims. ‘It was just a box I had to check.’
Many students wait tables to support their college expenses, Bertozzi played keyboard and sang backing vocals in a rock band called Bored of Education

Her realisation that chemistry interested her came via an organic chemistry class in her second year. ‘Its reputation was that the professors intentionally made it gruelling and difficult,’ she says. Her fellow students complained it was too much of a memory exercise but she was inspired. ‘It was by far the class that I enjoyed most and I didn’t find it at all tedious. It matched my brain.’
She changed her major from biology to chemistry a year later. Music didn’t completely fall by the wayside, though. Whereas many students wait tables to support their college expenses, Bertozzi played keyboard and sang backing vocals in a rock band. ‘It was the 1980s, and we were a heavy-metal hair band called Bored of Education.’ They were paid to play their mix of self-penned metal songs and pop covers at events such as college parties.
It also led to a brief brush with fame – lead guitarist Tom Morello went to Los Angeles after graduation and formed the band Rage Against the Machine. Bertozzi can’t help but wonder what might have been had she followed suit. ‘I didn’t get on that bus, and my playing is now limited to “The wheels on the bus go round and round”. I’m waiting for my sons to get old enough to appreciate 1980s heavy metal!’

Searching for an opportunity
Harvard science undergraduates are encouraged to work in research labs over the summer. ‘I tried to get a job in an organic chemistry lab after my [second] year but they all said they were full,’ she says. Instead, she wound up in a lab studying oxidative membrane damage at Massachusetts Eye and Ear hospital. She had already been working with the professor, Irene Kochevar, as a ‘gofer’ for a couple of hours a week during term-time, visiting the medical school library three miles away with a big bag of quarters to photocopy papers.
At that time, laser eye surgery was a new thing. ‘It was important to understand whether eye tissues would be damaged by surgical lasers,’ she says. It was a slightly chaotic experience – she helped make ‘ghosts’ from erythrocytes by osmotically shocking them to leave the membranes and then irradiating them with a laser to see if they would be damaged.
‘I started to get a feeling that maybe it wasn’t a good idea to be an organic chemist.’
Carolyn Betozzi
There was no organic synthesis job for her the next summer, either. ‘I took the first-year graduate course in physical organic chemistry and got to know some of the grad students. I’d visit their labs and thought they might introduce me to their PI [principal investigator], which could help me get a summer job,’ she says. But it didn’t. ‘I started to get a feeling that maybe it wasn’t a good idea to be an organic chemist.’
She lined up a job in a biochemistry lab instead but then met Joe Grabowski, an assistant professor in physical chemistry, who was teaching that graduate class. His lab had just one grad student – at that time, Harvard assistant professors found it hard to recruit grad students because of the uncertainty about their own long-term prospects. The success rate for gaining tenure was low – tenured professors were typically recruited from outside – leaving new academics to struggle along until they moved to another university.
Grabowski was open to recruiting undergrads instead. ‘One day after class he asked if I wanted to spend the summer in his lab,’ she says. ‘I was blown away that this opportunity landed in my lap, so I bailed on the “Plan B” biochemistry lab job.’

Figuring things out on the fly
Grabowski wanted to start a project in photoacoustic calorimetry, measuring pressure waves formed when photoexcited molecules relax and emit heat, causing a local solvent expansion. ‘You can put a microphone on the edge of the cuvette and hear the waves,’ she says. ‘The amplitude of the wave tells you something about the amount of energy that is dissipated as heat.’
’It was good experience to be handed a project that was way over my head, so I had to figure everything out on the fly’
Carolyn Bertozzi
Bertozzi claims it was all a little over her head. ‘At the beginning of the summer, I couldn’t see the path from me to the instrument,’ she says. ‘There are some things you can be sure will never happen – if I’m watching an NBA game I know for sure that there is no path between me and being a professional basketball player! But here there was supposed to be a path and, somehow, I had to get there.’
As well as building and assembling components to measure acoustic waves and digitise those analogue signals, she embarked on learning to program in a language called Forth so the instrument could be controlled. By the end of the summer she had created routines that transformed those digital signals into graphs.

She wrote her undergraduate thesis on the project and by the time she left the instrument had been built, although she never got to do any experiments with it. ‘Finally, as I was finishing my PhD, the paper came out with my name on it!’ she says. ‘It wasn’t organic synthesis but in retrospect it was very empowering. It taught me you don’t have to know the path to a big goal – every day you do something and the weeds slowly clear out, and the path becomes clearer over time. It’s easy to get spooked by a big challenge but you’re not supposed to do it in a day. It was good experience to be handed a project that was way over my head, so I had to figure everything out on the fly.’
She remained desperate to do organic synthesis, though. ‘As much as tinkering with computers and lasers was fun, I really liked thinking about how organic molecules behave and the notion of creating new ones, and I was inspired by a class taught by Jeremy Knowles in bioorganic chemistry,’ she says. ‘I was fascinated by how enzymes manipulated molecules.’
A summer internship at Bell Labs in New Jersey provided further inspiration. She worked with Christopher Chidsey, a young surface scientist studying self-assembled monolayers of alkane thiols on gold surfaces. ‘I still didn’t do any synthesis but at least I got to dissolve and prepare organic compounds!’ she says. Chidsey was bemoaning the absence of decent software to manage the data from the cyclic voltammeter and was contemplating using Forth. ‘I felt like I had a useful skill for a change!’ she laughs. The Journal of the American Chemical Society paper that came out of that summer remains one of her most highly cited papers, despite all the influential work she has published since.
For two years, Bertozzi and two grad student labmates ran their own operation, carrying out research and submitting papers to journals (and even calling the editors to complain about reviews)
A synthetic chemist at last
Her dream of synthesis was finally realised when she arrived at the University of California, Berkeley, for grad school, designing molecules for studying biology in the lab of Mark Bednarski, a young academic she had met at Harvard when he was a postdoc in George Whitesides’ lab. He was interested in carbohydrate chemistry and this was about the time the neuraminidase inhibitor flu drugs Relenza (zanamivir) and Tamiflu (oseltamivir) were being developed, both of which resembled the sugar sialic acid. ‘The structures of flu neuraminidase and haemagglutinin had just had been solved, and it was an exciting area to dive into,’ she says.
But in her third year, Bednarski was diagnosed with colon cancer. He took a leave of absence for treatment, then decided that he wanted to be a doctor and enrolled as a medical student at Stanford University. Rather than moving to another group, Bertozzi and a couple of labmates went it alone, and finished their PhDs without supervision. ‘This wouldn’t happen now because of regulation and safety concerns but back then it was a bit of a Wild West!’ she recalls. ‘I was grateful because my project was finally working and I didn’t want to start all over again in a new lab.’
For two years, Bertozzi and two grad student labmates ran their own operation, carrying out research and submitting papers to journals (and even calling the editors to complain about reviews). ‘We didn’t know how else to do it,’ she says. ‘At the time we were a bit frustrated but, in retrospect, it demystified the job of PI and I learnt early on what needs to be done to run a lab, albeit a small one.’
During a subsequent postdoc in an immunology lab at the University of California, San Francisco (UCSF), she met up with Chidsey again, who by then was on the faculty at Stanford. He encouraged her to apply for an academic job as a chemical biologist, which Stanford was searching for at the time. ‘I realised I wanted to stay in the Bay area and also applied to Berkeley and UCSF,’ she recalls. All three gave her offers and she chose to return to Berkeley.

The birth of bioorthogonal chemistry
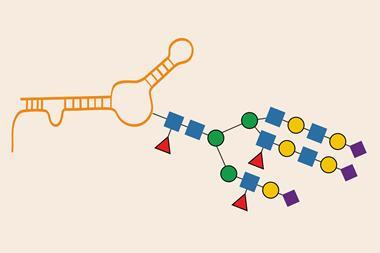
Her postdoc had made Bertozzi realise just how frustrating it was to study glycoscience in biological systems. ‘The tools were so primitive, there was no way to take measurements on cells, organisms or tissues, and I thought there had to be a better way,’ she says. ‘It opened my eyes to what was missing in the technology space.’
That became her initial scientific mission back at Berkeley: developing tools that enable glycobiologists to make faster, broader progress. Unlike for proteins or nucleic acides, there was no imaging technology for sugars. So, she set about developing an imaging tool to put probes onto sugars so they could be monitored. This led to metabolic labelling methods – inserting labels into sugars via metabolic precursors in the diet of the cells that make them. ‘The probes themselves cannot be inserted in this way as they are too large and the enzymes will destroy them but you could put a chemical marker on the sugar and then add probes afterwards,’ she says.
’I remember thinking: “Why not? If I can build a photoacoustic calorimeter as an undergrad, why shouldn’t a grad student work on TB?”’
Carolyn Bertozzi
This mission led Bertozzi to develop new types of reactions she describes as ‘bioorthogonal chemistry’, chemical reactions that occur inside living things without interacting or interfering with any native biological processes. Part of her lab was developing bioorthogonal reaction methods and the rest carried out applications and imaging with the labelled sugars they had created.
‘New ideas then started spinning out of ongoing projects,’ she says. One was in tuberculosis (TB) – the bacterial genome had just been sequenced and grad student Joseph Mougos (now a successful academic in his own right) was keen to look into it. ‘We were already studying various human enzymes, so we blasted their gene sequences against the TB genome to see if we could discover microbial relatives,’ she says. ‘To our surprise, he found distant relatives of human sulfotransferases, which put sulfate groups onto sugars.’
The next step was to make knock-out mutants and look for phenotypes, but this was easier said than done, as TB requires a biosafety level 3 containment facility, which Bertozzi didn’t have. ‘I was an assistant professor, on a tight budget, but I remember thinking: “Why not? If I can build a photoacoustic calorimeter as an undergrad, why shouldn’t a grad student work on TB?” Joseph found the necessary containment facility, figured out how to do the mutagenesis, and we developed a mass spec platform that allowed us to use sulfur-34-enriched heavy sulfate labelling to look for metabolites the enzymes might have made.’
Having figured out the biosynthetic pathway of a sulfated glycolipid believed to be a virulence factor, they realised this metabolic process could be applied to imaging. Even more excitingly, it became clear these imaging tools might detect TB cells in an infected patient’s sputum, using just a battery-powered microscope. These instruments are already widely used to diagnose malaria, and it could represent an important advance in TB diagnosis in low-resource environments where the disease is endemic.
‘We created a fluorogenic labelling reagent, and this project is now supported by the Bill and Melinda Gates Foundation to create a point-of-care diagnostic,’ Bertozzi says. ‘We would be nowhere near this if, all those years ago, one student hadn’t pushed to work on TB, and we built the path over a decade, one experiment at a time.’
Moving across the bay
After almost 20 years at Berkeley, Stanford came calling again in 2015. ‘This time the stars had aligned, and a big investment was being made in chemical biology, with a new building and 20 new faculty members, including a couple of senior professors,’ she says. Stanford was building a new institute, ChEM-H (Chemistry, Engineering and Medicine for Human Health), and she saw an opportunity to contribute from the ground up.
‘I wanted my students to gain experience in the ecosystem of medicine’
Carolyn Bertozzi
Bertozzi hadn’t been looking to leave Berkeley, and the timing wasn’t ideal as she and her partner were about to have their third baby, but she knew immediately she wanted the job. ‘I wanted my students to gain experience in the ecosystem of medicine,’ she says. ‘That wasn’t easy at Berkeley, but Stanford has a med school, so we made the move to Palo Alto.’
The next unexpected research direction turned up almost immediately. ‘I had a cold call from a Stanford alumnus, Matt Wilsey, who was a successful tech entrepreneur in Silicon Valley,’ she says. ‘His young daughter Grace had been diagnosed with an ultra-rare genetic disorder: she had two faulty versions of the NGly1 gene, which encodes for an enzyme called N-glycanase that removes sugars from misfolded glycoproteins in advance of their degradation. He had set up a non-profit research foundation, with the goal of finding treatments and a cure. Given my lab’s focus in glycoscience, he wondered if I could contribute.’

Wilsey had already recruited several high profile geneticists and biologists but no chemists with a focus on sugars. ‘I was hesitant to put forth a research proposal without a better understanding of how loss of the N-glycanase enzyme was making these children sick,’ she says. The function of this enzyme was not well understood at the time and there was no good hypothesis to link its function to patient symptoms.
The months went by and another coincidence came along. At a meeting, Bertozzi got chatting to Caltech biologist Ray Deshaies, who has since moved to Amgen. N-Glycanase nestles close to the proteasome, so it was assumed its role was to remove large glycans before the proteasome degraded misfolded proteins into small peptide fragments. Yet cultured cells engineered with the same NGly1 mutations weren’t filling up with undigested glycoprotein ‘garbage’ in the way one might expect – cells in a lab dish were mostly fine without their N-glycanase, while both humans and laboratory mice became very sick. Something was clearly missing.
The research community had started to suspect that important substrates are present in some cells, from which sugar units must be removed by N-glycanase if a person is to remain healthy. But finding those substrates was a major challenge.
‘I told Deshaies this story, and it turns out he was the right person to ask,’ she says. A few years earlier, he had worked on an unusual transcription factor, Nrf1, and he thought removal of N-glycans might be critical to its activation. Nrf1 drives the expression of proteasome subunit genes under conditions of proteasome insufficiency, such as when it reaches capacity or is inhibited by a drug. Deshaies was interested because of its potential role in resistance against proteasome inhibitor cancer drugs, one of which, carfilzomib, he had helped develop.
Based on that tip, Bertozzi wondered if removal of N-glycans by N-glycanase was an important step in the processing and activation of Nrf1. ‘For the whole rest of the meeting I sat in the back row googling this transcription factor and found reports about the phenotypes of various mouse knockouts – they essentially phenocopied the symptoms of the human NGly1 disease,’ she says. ‘I couldn’t believe it – this transcription factor was likely one of the most important substrates for the enzyme and as soon as I got back to Stanford I wrote a proposal.’
‘This is a project that started with a patient and her family, and now represents a major effort in my lab.’
Carolyn Bertozzi
A year on, it’s clear this supposition was completely on the money – both knock-out and patient-derived cell lines are very sensitive to proteasome inhibitors and the transcription factor is non-functioning in them. This deficiency, Bertozzi suspects, is likely responsible for several neurological symptoms experienced by NGly1 patients as, without active Nrf1, their cells cannot correct situations of proteasome insufficiency. Neurons are known to be particularly sensitive to problems with protein degradation and turnover.
‘My goal of bringing our research closer to clinical sciences at Stanford took less than a month,’ she says. ‘This is a project that started with a patient and her family, and now represents a major effort in my lab.’
Another growing interest of Bertozzi’s is the hot field of immuno-oncology, where glycosylation plays an important role in immunomodulation in the tumour microenvironment. ‘It’s a great time for glycoscientists – biologists are discovering important roles for glyco genes using Crispr-based genome screens and they are keen to work with experts who can figure out the relevant biochemical pathways. So if you’re the local glyco expert, lots of people call you to help explain their gene lists of glyco alphabet soup,’ she says.
‘Opportunities drive by you and if you are lucky you notice a few. We probably miss a lot as we cannot pay attention to every little thing and only have so much bandwidth. But opportunities are always driving by, and if you see it, you should get on the bus and see where the path takes you.’
Sarah Houlton is a science writer based in Hertfordshire, UK









No comments yet