AlphaFold has predicted the structures of almost every known protein
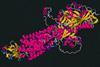
Google offshoot DeepMind has released more than 200 million predicted 3D structures, covering nearly the entire protein universe
The artificial intelligence company DeepMind, in partnership with the European Molecular Biology Laboratory (EMBL), has released the predicted structures for almost every protein known to science – over 200 million structures in total.
The structures were produced using DeepMind’s AlphaFold system, which predicts the 3D structure of proteins based on their amino acid sequences. The structures have been added to the open source AlphaFold Protein Structure Database, maintained by the EMBL. ‘It’s our gift to humanity, and a demonstration of the benefits AI can bring to society,’ announced DeepMind’s co-founder and chief executive Demis Hassabis on Twitter.