Scientists have found further evidence for the existence of an elusive intermediate implicated in chemical reactions that degrade atmospheric pollutants. A new method of directly detecting the simplest form of this intermediate, as well as more measurements of the intermediate’s reactivity, provide indications that atmospheric models need to improve how they account for them.
More than 50 years ago, German chemist Rudolf Criegee proposed that alkenes degrade by reacting with ozone to form a cyclic ozonide. One of the products formed when this ozonide falls apart is a carbonyl oxide called a Criegee intermediate.
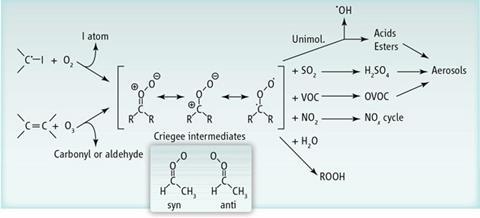
Two breakthroughs in detecting Criegee intermediates came last year. Craig Taatjes, at Sandia National Laboratory, US, and colleagues reported the first measurements of the reactivity of the simplest Criegee intermediate, CH2OO, using photoionisation mass spectrometry.1 While this method can also detect the intermediates, it requires specialised equipment, namely a synchrotron to make a photon beam to ionise the sample. Directly detecting the same molecule based on its absorption of ultraviolet light provides a stronger signal using more common instruments.2
Now, Yuan-Pern Lee and Henryk Witek, both at National Chiao Tung University in Taiwan, and colleagues have detected the simplest Criegee intermediate in the gas phase using another accessible technique: time-resolved Fourier-transform infrared absorption spectroscopy.3 This method uses bond vibrations to identify particular molecular structures, in this case the carbonyl oxide unique to Criegee intermediates. With a simpler way to identify the molecular fingerprint of these intermediates, scientists might be able to monitor their reactions with other molecules, the researchers say.
Structural speed-up
New measurements of the reactivity of the second heaviest Criegee intermediate, acetaldehyde oxide (CH3CHOO), reveal that its structure influences its reactivity.4 This intermediate can exist in one of two conformers. The syn conformer has the methyl group and the second oxygen on the same side of the molecule, while the groups are opposite each other in the anti conformer.
Taatjes and Carl Percival, at the University of Manchester, UK, realised that their previous method of detecting Criegee intermediates by photoionisation could also identify each conformer of acetaldehyde oxide. The researchers then measured the reactivity of this intermediate with water, NO2 and SO2. They found that the anti conformer reacts faster with water and SO2 than the syn conformer does.
Knowing how quickly one Criegee conformer reacts with water can help scientists determine how long these intermediates exist in the atmosphere, Taatjes says. The kinetics of the reaction with SO2, now known to be orders of magnitude faster than predicted by models, combined with the direct detection of sulfuric acid as a product indicates that Criegee intermediates might influence other processes in the atmosphere. Sulfuric acid can seed sulfate aerosols, which reflect light away from the planet. The next step is to determine the amount of sulfate aerosols produced by Criegee intermediates and figure out if those aerosols affect climate, Percival says.
Given the differences between the predicted and measured reaction rates even for the smallest Criegee intermediates, it’s hard to know how structural differences will alter the reactivity of other molecules in this family of intermediates, says Marsha Lester, of the University of Pennsylvania, US. ‘There will still be more surprises.’
‘The final quantification of Criegee intermediate chemistry is still a long way off, but the results we have so far strongly suggest we will have to adjust the atmospheric chemical models to account for Criegee intermediates,’ says Luc Vereecken, of the Max Plank Institute for Chemistry.
References
1 O Welz et al, Science, 2012, 335, 204 (DOI: 10.1126/science.1213229)
2 J Beames et al, J. Am. Chem. Soc., 2012, 134, 20045 (DOI: 10.1021/ja310603j)
3 Y-T Su et al, Science, 2013, 340, 174 (DOI: 10.1126/science.1234369)
4 C Taatjes et al, Science, 2013, 340, 177 (DOI: 10.1126/science.1234689)






No comments yet