Scientists discover a simple way to form carbon–carbon bonds from carbon dioxide without using synthetic or biological catalysts
Molten salts containing alkali metal cations can be used to generate species that easily react with carbon dioxide to produce organic compounds. The approach could enable the synthesis of useful chemicals for plastic production from carbon dioxide without any highly reactive reagents.
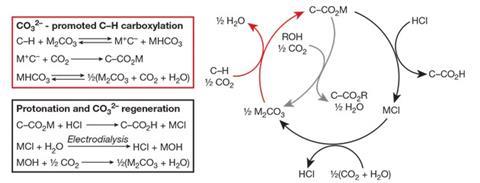
Using carbon dioxide as a raw material for commodity synthesis would be an excellent way to convert greenhouse gas emissions into valuable products, but unfortunately it’s exceptionally tricky. Although carbon dioxide has been used by the chemical industry, for example to produce urea, it is relatively unreactive so that specific conditions including acidic carbon–hydrogen bonds or high energy reagents are usually required.
Inspired by plants’ carbon–carbon bond formation, US scientists have now found a way to circumvent this problem. ‘Our method works by simple acid–base chemistry,’ says Matthew Kanan from Stanford University, who led the study. He and his colleagues used carboxylate salts as the reaction medium. ‘These compounds melt at 200 to 350°C to form a molten salt,’ Kanan says. ‘In this medium, we found that carbonate is able to deprotonate C–H bonds that are ordinarily extremely weak acids.’ Kanan says that the carbanion intermediate formed is able to react with carbon dioxide to form a carboxylate. By combining this simple carboxylation reaction with a hydrogenation process, it is possible to synthesise a variety of products.
‘The idea to use molten salts in this reaction is really visionary and will undoubtedly lead to even more opportunities to generate reactive species on scale and incorporate carbon dioxide into even more processes,’ says Cathleen Crudden, who leads two catalysis groups at Queens University, Canada, and the Institute of Transformative Bio-Molecules in Japan. The presence of the molten salt is essential for the chemistry, says Kanan. He explains that the acid–base properties of organic compounds in molten salts are very different from those in conventional solutions, and enables reactions that would not be possible in other media. ‘I don’t think researchers previously appreciated the remarkable acid–base reactions that are possible in a molten salt with alkali cations,’ he says.
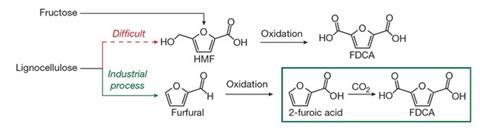
To demonstrate a possible application of their method, the team synthesised furan-2,5-dicarboxylic acid (FDCA), which can be used to produce polyethylene furandicarboxylate (PEF). ‘PEF is a possible replacement for petroleum-derived polyethylene terephthalate (PET), which is a very large scale polymer that is currently made from non-renewable sources,’ says Kanan. One of the starting materials for producing FDCA is non-food biomass, and the carboxylate salt used in the reaction can be recycled too. The results indicate that it might be possible to produce PET from carbon dioxide as well.
‘The use of carbon dioxide to generate liquid fuels is an extremely important topic,’ says Crudden. ‘However, when reasonably high value polymers that are produced in large scale – such as the ones described herein – are targeted, this is also a very valuable use of carbon dioxide. In addition to finding a use for captured carbon dioxide, this concept also decreases our reliance on petroleum as a starting material.’
References
A Banerjee et al, Nature, 2016, DOI: 10.1038/nature17185











No comments yet