Japanese researchers have discovered that the chirality of one molecule can be sensed from a change in shape of another.
Takanori Suzuki and colleagues at Hokkaido University, Japan, have discovered a clever host molecule whose change in shape can reveal the chirality of guest molecules bound to it.
The ability of molecules to chemically recognise each other is fundamental in biochemistry. In particular, the shape of a guest biomolecule is often its unique molecular key that fits the lock of a matching host. This recognition then results in specific biochemical behaviour when they bind.
Inspired by this principle, Suzuki made a series of macrocyclic molecules containing a large ring that included eight para-phenyl groups and two alkyne linkages. These host molecules also contained two pairs of amide groups on opposite sides of the macrocyclic ring. This allowed two guests to bind, or chelate, to it at the same time.
The crystal structures of these hosts showed that their molecules have a rectangular shape in the solid state, but when they are chelated by two chiral S,S-bis(ammonium) guests, they become twisted into a figure-of-eight. This effect and the direction of the twisting was revealed by the circular dichroism spectrum of the complex.
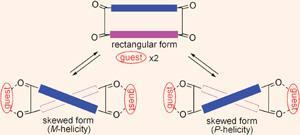
However, Suzuki’s most important findings came when he compared the behaviour of R,R,R,R- and S,S,S,S-enantiomer pairs of a chiral host with the same S,S-bis(ammonium) guest. The twisting of the host only took place when the chirality of the host and guests matched - for example when a S,S,S,S -host was bound by a pair of S,S-guests. In contrast, for a mismatched combination, like a R,R,R,R-host and S,S-guests, the rectangular shape remained.
This ’stereospecific chiroptical modulation’, as Suzuki termed it, is a striking and novel example of chiral recognition in action. This type of recognition has a strong potential for use in biochemical sensors of the future.
David J Parker
References
R Katoono, H Kawai, K Fujiwara and T Suzuki, Chem. Commun., 2005, (DOI: 10.1039/<MAN>b510134d</MAN>)






No comments yet