New microwave instrument captures rearranging isomers
A new microwave spectrometer has allowed US scientists to track molecules writhing through different geometric shapes when excited - opening a new window on their reactivity. The first published experiments show that one molecule’s conversion between isomeric forms may be slower than theory predicts.
When a molecule has sufficient energy it can switch between various atomic arrangements or stereoisomers, with the interconversion sometimes taking place in a matter of picoseconds. The different isomers are identified by their unique rotational spectra - they absorb different frequencies of microwave radiation. But watching the shape changes take place requires capturing high-resolution spectra extremely quickly.
Brian Dian and colleagues at the University of Virginia have achieved this with a chirped-pulse Fourier transform microwave (CP-FTMW) spectrometer, which captures spectra in a single measurement at least 100 times faster than conventional microwave instruments.
The new instrument bombards a molecule that has been excited by a laser with microwaves covering a wide range of frequencies. ’This spectrometer has made it possible to measure the broadband rotational spectra following laser excitation in about 10 seconds,’ says co-author Brooks Pate.
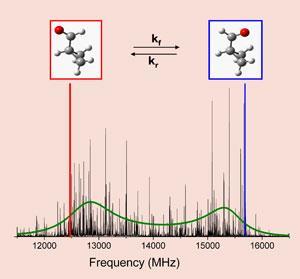
To explore how a molecule moves between stereoisomers when excited, many rotational spectra are acquired at a range of excitation levels. Using the new spectrometer, the researchers performed 17,000 rotational scans in 52 hours. A traditional cavity-FTMW spectrometer would require 27 years to complete the same measurements.
The reduced measurement time allowed the researchers to examine how a cyclopropane carboxaldehyde molecule rearranges between its syn and anti stereoisomers. The findings revealed complex isomerisation kinetics and, surprisingly, that the rate of isomerisation is significantly slower (by a factor of 16) than predicted by theory.
The researchers believe the technique offers several advantages, especially in the acquisition of the full spectral range of samples. ’The spectrometer ... has potential applications in analytical chemistry, biomolecule spectroscopy, and the detection and characterisation of reactive species,’ says Pate.
’The CP-FTMW spectrometer built by Pate and co-workers [is] a very important instrumental advance,’ says spectroscopist Terry Miller of Ohio State University. ’It very significantly improves the sensitivity and reduces the time required to take a significant portion of the microwave spectrum of a molecule.’
Cordelia Sealy
Enjoy this story? Spread the word using the ’tools’ menu on the left.
References
et al, Science320, 924 (DOI: 10.1126/science.1157668






No comments yet