Nanosilver is filtering into the environment in ever-increasing quantities. But is it the nano or the silver component we should be worrying about, asks Hayley Birch
Nanosilver is filtering into the environment in ever-increasing quantities. But is it the nano or the silver component we should be worrying about, asks Hayley Birch
Worms are discerning when it comes to soil. Soil, you might say, is their area of expertise. They can sense if they’re wriggling into the wrong sort of soil - one that might contain pesticides or heavy metals - and adjust their trajectory accordingly. Their talents are held in such high regard that the International Organization for Standardization (ISO) has seen fit to give the earthworm its own internationally recognised test standard for determining the chemical quality of soils.

Paul Bertsch at the University of Kentucky’s college of agriculture in Lexington, US, knows their abilities only too well. In his lab, many hours have been devoted to observing the worms as they make their deliberations. And Bertsch and his team have recently discovered that the earthworm’s powers of perception extend all the way down to the nanoscale.

The ISO standard test places a clew (the collective noun) of worms into a sort of choice chamber, between a control soil on one side and a contaminated test soil on the other. Bertsch’s team found that earthworms behave in a different way faced with ordinary dissolved silver from silver nitrate (AgNO3) compared to soil contaminated with silver nanoparticles.1 ’They avoid the soil that has the dissolved silver in it immediately, whereas with the nanoparticles it takes some time for them to intercept some of these particles and then decide that this isn’t a good environment,’ says Bertsch. ’And then they leave.’
Although the sensory mechanisms involved are unknown, the really interesting aspect, according to Bertsch, is the change in the animals’ behaviour. What it means in broader terms, however, is hard to say. Researchers are still grappling with the complex pathways and effects of silver nanoparticles entering the environment. But as they are used in an ever-increasing number of products - from wound dressings to water filters - the opportunity to avoid any unforeseen consequences is slipping away.
Sizing up the issue
Just getting a handle on the scale of the problem is tricky, especially given that current requirements for declaring nano-ingredients in products are hazy. This is now starting to change; the US Environmental Protection Agency recently sought comments on proposed approaches to registering pesticides containing nanoscale materials, while in the EU there have been calls for a clearer definition of nanomaterials within policy making.
As of March 2011, a nanotechnology consumer products inventory (established by the US-based Pew Charitable Trusts and Woodrow Wilson International Center for Scholars) listed over 1300 products containing nanomaterials, from 30 different countries. Some of the most commonly listed nano-ingredients are metals, including titanium, zinc and gold. Nanosilver sits right at the top of the list, showing rapid growth from less than 30 products in 2006 to over 300 in 2011.
Of the nano-form metals already finding their way from consumer products into water systems and soils, silver - due to its widespread use and toxicity - is the one that has faced the most intense scrutiny. Its major application is as an antibacterial, meaning it is primed to snuff out life. As Bertsch points out, although ill effects are only seen in humans at very high concentrations, silver itself is well known for its toxicity to other organisms, including soil and aquatic species. This was a bigger issue when the photographic industry was at its peak, spewing out silver in film coatings. As yet, though, it is unclear whether the toxic effects of nanosilver are related to the small size of the particles, or the release of silver ions, as in ordinary silver compounds.
Coming out in the wash

Either way, nanosilver should only become a concern if sufficiently large quantities are being released from consumer products. Bernd Nowack, who studies the environmental behaviour of nanomaterials at the Empa Technology and Society Laboratory in St Gallen, Switzerland, has been exploring what he considers a ’worst case scenario’ - the rough and tumble, and high temperature chemistry of the domestic washing machine. His 2009 study on washing of antibacterial textiles, including socks, containing nanosilver suggested that up to a third of the nanoparticles they contained could be released in the first wash.2 But particles that were bound loosely at the surface of the fabric were lost far more easily. ’It matters a lot how the particles are incorporated into the product,’ says Nowack. ’Where they’re incorporated into the matrix, there’s much less release. I think from this study we cannot make any guesses about how it is with completely different products.’
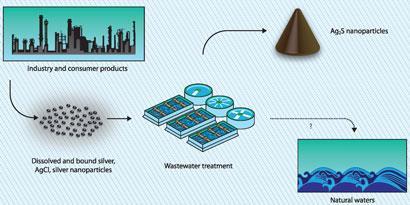
All this uncertainty makes it impossible to predict how much nanoparticulate matter might be entering our sewers. A study commisioned by the UK Department for Environment, Food and Rural Affairs (Defra), published in April 2011, found an average figure of 6ng/l - far less than predicted by consumption data - for colloidal silver (2-450nm) in water leaving sewage works in England and Wales. Colloidal silver made up 1% of silver entering and 7% of silver leaving sewage treatment plants. However, it is unclear whether the smaller particles (below 100nm) ordinarily defined as ’nano’ might be more, or less, easily retained than the larger ones. According to the study, 97% of total silver, in all forms, is removed during wastewater treatment. Which begs the question: where does it end up?
Sludge, says Stephen Lofts, who is based at the Centre for Ecology and Hydrology in Lancaster, UK. Lofts has been working on an EU-funded project called Nanofate, which is exploring the environmental fates of nanoparticles. ’The main question we’ve been trying to deal with in the first 18 months is where do they go if they hit the sewage works? Do they go into the sludge and onto the land or do they go straight through and into the river?’ says Lofts. ’The evidence so far suggests that the main portion will end up going into the sludge and onto the land.’ Lofts is also collaborating with Bertsch in a UK-US consortium. The Transatlantic Initiative for Nanotechnology and the Environment is developing a pilot scale wastewater treatment plant that will produce sludge from water contaminated with nanoparticles at realistic doses. Silver and other commercially relevant nanoparticles, including titanium dioxide (a component in some sunscreens), will feature heavily in the experiments.
Tarnished by time
In some countries - such as Switzerland - the spreading of sewage sludge on farmland is banned, whereas elsewhere any silver nanoparticles in sludge are going straight onto the soil in which our food grows. But by this point they may no longer resemble the pristine, as-manufactured nanoparticles that most researchers are using to try to understand toxicity in the lab, raising questions about the relevance of these experiments.
Normal non-nano silver dissolves to produce toxic silver ions. According to Lofts, once released into the environment, silver and copper (and some other metal) nanoparticles should, to varying degrees, do the same. In the case of nanosilver, by the time it reaches the treatment plant, much of it will already have been snapped up by sulfur and incorporated into sulfides. Robert Hurt at Brown University, Rhode Island, in the US, and colleagues showed silver nanoparticles react with dissolved sulfide species present in wastewater to form silver sulfide nanostructures akin to those emerging from sewage treatment.3
Back at Kentucky University, Bertsch’s group has been trying to work out what happens to the structure of copper and silver nanoparticles over time - what happens as they ’age’. Copper nanoparticles are used in fuel cell catalysts and engine coolants, as well as pesticides and fungicides. Through close inspection of the electronic structure of copper nanoparticles using absorption spectroscopy techniques, Bertsch’s team found that they rapidly oxidised in air.4 ’We get copper oxide that forms at the surface,’ says Bertsch. ’It appears that the copper metal stays in the core and we see initial oxidation and reactivity, but after 28 days in the soil we don’t really see any further ageing.’
Similar reactivity has been noted for silver nanoparticles, with the outer layer of oxidised metal developing more slowly than for copper. The silver ions produced when these outer, oxidised layers dissolve are often blamed for nanosilver’s toxic effects. In Bertsch’s experiments with earthworms, silver ions from AgNO3 have proved more toxic than aged silver nanoparticles.5 To achieve the same toxicity with nanoparticles, silver concentrations 10 times higher than those for ionic silver were needed - and it’s possible the toxic effects of the nanoparticles were also related to free silver ions, produced during ageing. On the other hand, the separate experiments in which the worms avoided nanoparticles in a markedly different way to ionic silver suggest there is some specifically nano-related response going on.

James Hutchison, a materials chemist at the University of Oregon, in Eugene, US, says too little thought is given to the complex reactions of silver in the environment. ’Silver in general is a very dynamic system, where the speciation is complicated by the fact that you can have bulk silver and nanoparticle silver ions all participating and all having their own states,’ says Hutchison. From a regulatory point of view, he adds, it might be better to think about the total amount of silver in the environment rather than the nanoparticle fraction more specifically. This kind of thinking is predicated on his team’s study published in October 2011, which highlights a point that is very often forgotten: that not all nanoparticles are manmade.6 In fact, metallic nanoparticles may have been around for a very long time indeed.
The Oregon group’s study reveals that metal objects including silver spoons and copper wire naturally yield metal nanoparticles when exposed to moisture - a finding that so surprised one of the reviewers of the paper, says Hutchison, that at first they wouldn’t believe it. This realisation, and communications from other scientists suggesting that the phenomenon occurs more broadly than just for silver and copper, has led some to reconsider nanoparticles as materials that nature has been dealing with for millions of years.
Nano evolution
On the other hand, Hutchison’s study is far from the first to raise questions about the novelty of manufactured nanoparticles. A policy analysis by Nowack highlights papers detailing medical uses of nanosilver going back to the early 20th century.7 But even more relevant to today are studies by Bojeong Kim and colleagues at Virginia Tech, in Blacksburg, US, which suggested that wastewater treatment plants are efficient at transforming any type of silver into nanosilver sulfides. The implication being that sewage works have always churned out nanosilver in sludge - regardless of the quantities going in - and potentially in huge volumes prior to the collapse of the photographic industry. This certainly puts a new spin on wastewater figures, as the Defra study only accounted for total inputs and outputs at treatment plants, without considering how much nanosilver might be formed somewhere in between.
There is so much of the stuff floating about in the environment that Nowack says even banning nanosilver entirely wouldn’t make any difference. ’People think that if they ban nanosilver there’s no silver problem, but banning nanosilver doesn’t change anything,’ he says. ’If you maybe take away 5% of the silver flow, you still have 95% of silver ending up in sludge and on soils, just from some other sources.’ He also argues that much of the work on silver ion toxicity is irrelevant because silver in the environment is rarely found as free ions - it is usually bound up in compounds so that it is not in a form that is bioavailable. The silver sulfides pouring out of waste treatment plants are insoluble and, according to Nowack, pose no real threat.
The idea that we and all other species have evolved with these naturally occurring nanomaterials is key, says Bertsch. Not to mention that his worms’ uncanny avoidance of silver nanoparticles now looks more like a normal response that has simply gone unnoticed. ’The question is: what, if anything, is fundamentally different with manufactured nanomaterials?’ asks Bertsch. One answer is the coatings or surface modifiers that the various manufacturers of nanoparticles use, which may affect their bioavailability and toxicity. For example, San Diego-based company nanoComposix manufactures silica coated silver nanoparticles - the silica shell increases the stability of the particles in different solvents.
Pondering what happens as these manufactured materials continue to accumulate in water and soils, Bertsch hints that the final fate of silver nanoparticles - in all their commercial formulations - is still sketchy. ’What are they going to look like as they go through that ageing process over a longer term?’ he asks. ’Are they totally benign or do they still pose a hazard? Those are the things that a lot of people will be looking at over the next several years.’
Hayley Birch is a science writer based in Bristol, UK
References
1 W A Shoults-Wilson et al, Ecotoxicology, 2011, 20, 385 (DOI: 10.1007/10646-010-0590-0)
2 L Geranio, M Heuberger and B Nowack, Environ. Sci. Technol., 2009, 43, 8113 (DOI: 10.1021/es9018332)
3 J Liu, K G Pennell and R H Hurt, Environ. Sci. Technol., 2011, 45, 7345 (DOI: 10.1021/es201539s)
4 J M Unrine et al, J. Environ. Qual., 2010, 39, 1942 (DOI: 10.2134/jeq2009.0387)
5 W A Shoults-Wilson et al, Soil Sci. Soc. Am. J., 2011, 75, 365 (DOI: 10.2136/sssaj2010.0127nps)
6 R D Glover, J M Miller and J E Hutchison, ACS Nano, 2011, 5, 8950 (DOI: 10.1021/nn2031319)
7 B Nowack, H F Krug and M Height, Environ. Sci. Technol., 2011, 45, 1177 (DOI: 10.1021/es103316q)






No comments yet